Synthesis and application of mixed manganese - Iron oxide nanoparticles for adsorption of as(v) from aqueous solutions
A simple method has been used to synthesize nanoparticles of mixed manganese-iron oxide
for the adsorption of As(V) metal ions from aqueous solutions. Transmission Electron
Microscopy (TEM), X-Ray diffraction (XRD), Scanning Electron Microscopy (SEM), Fourier
transform infrared spectroscopy (FTIR), BET analysis were used to determine particle size
and characterization of produced nanoparticles. The x-ray diffraction pattern indicated that
the as-synthesized adsorbent is amorphous with 288.268 m2/g surface area; the amorphous
synthesized products were aggregated with many nanosized particles. The crystallinity of the
Mn2O3/Fe2O3 were obtained at 400°C and 600°C calcination temperature. The FTIR spectra
confirmed the presence of -OH group and H-O-H group localized at 3200 - 3400 cm–1 and
1618-1653 cm−1; theses intense bands is weak (fade) at the high calcination temperature of
the mixed manganese-iron oxide nanoparticles. In addition, when the calcination
temperature of the mixed manganese-iron oxide nanoparticles was 400OC, the weak
absorption bands at 630 cm−1 due to the vibrations of (Fe-O). The results showed that the
mixed manganese-iron oxide nanoparticles has high selectivity for As(V).

Trang 1

Trang 2

Trang 3

Trang 4

Trang 5
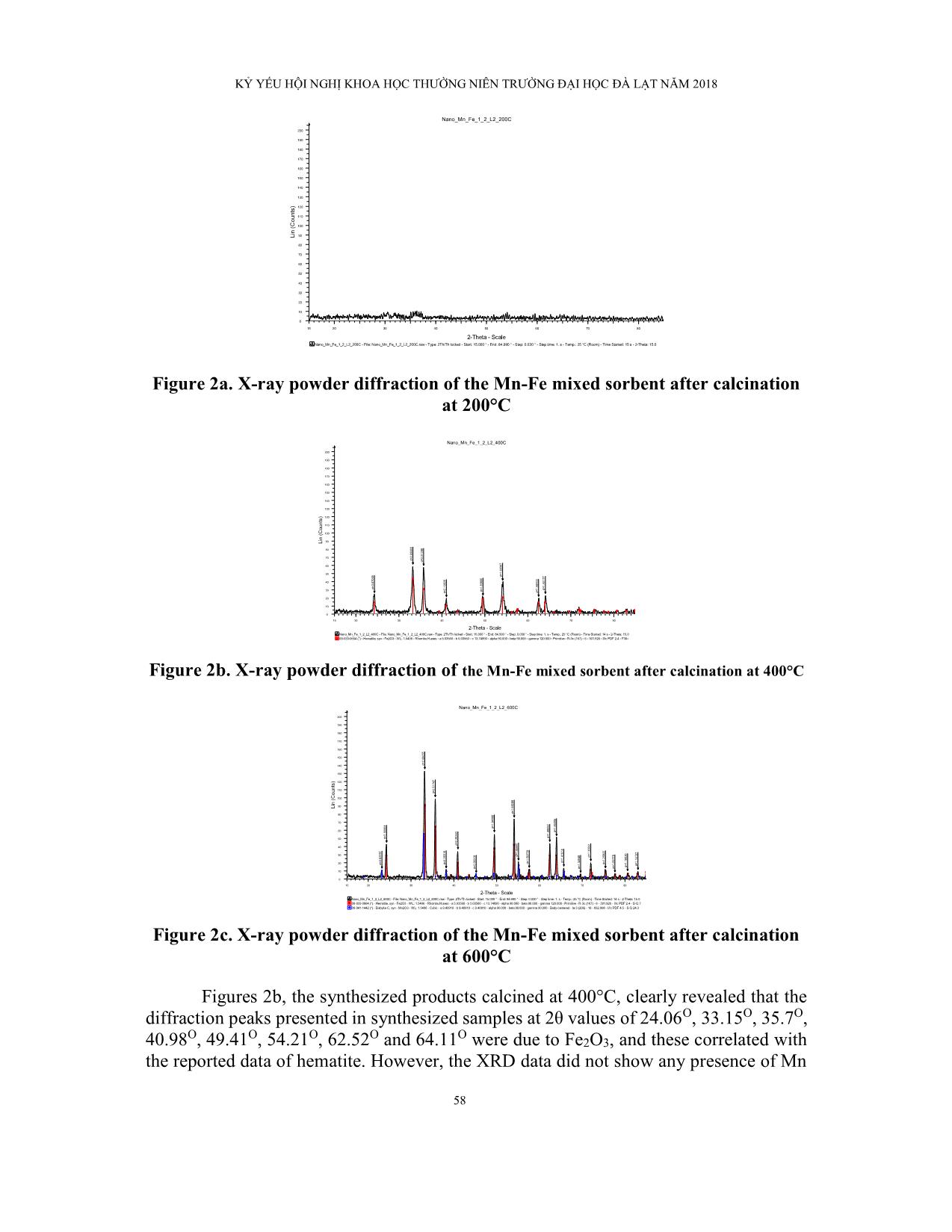
Trang 6
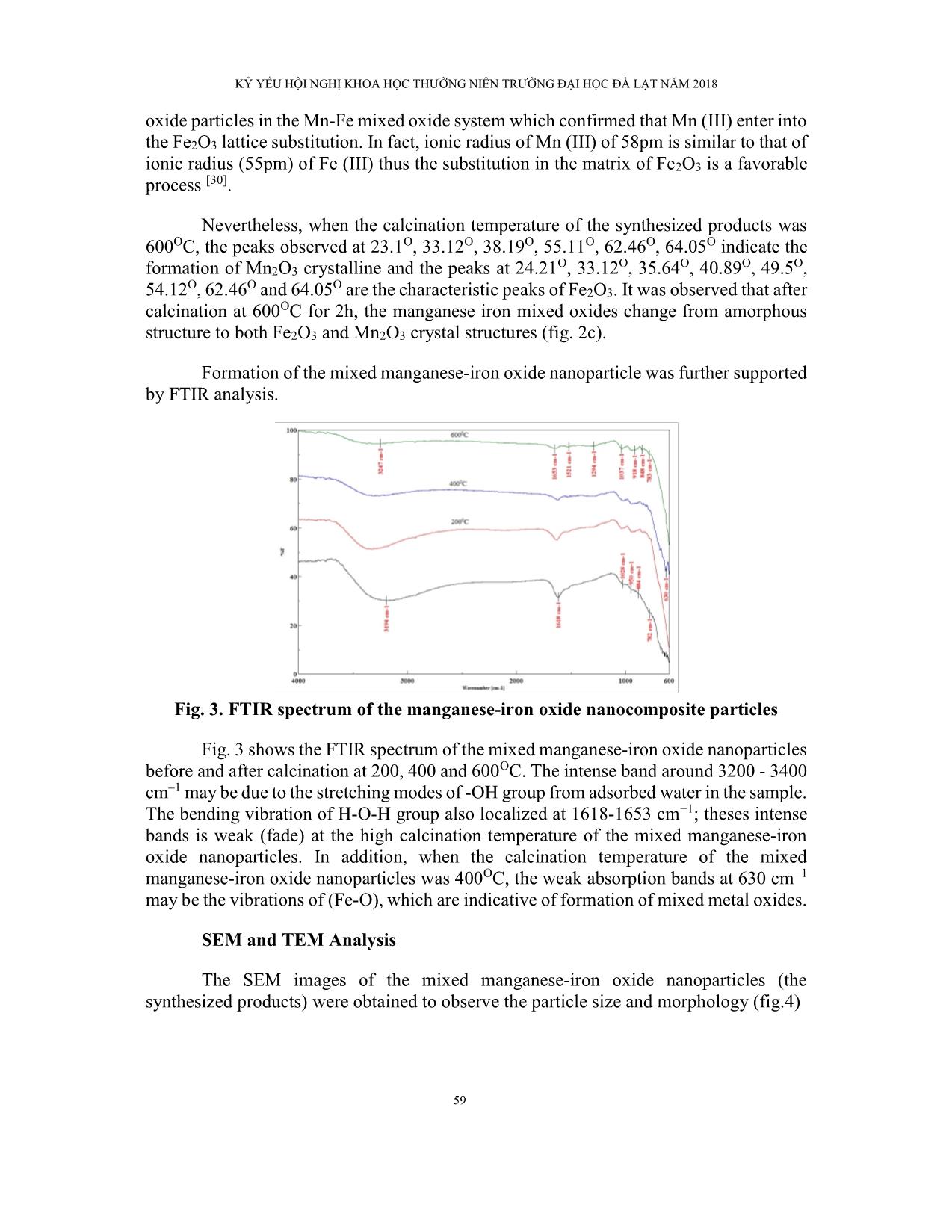
Trang 7
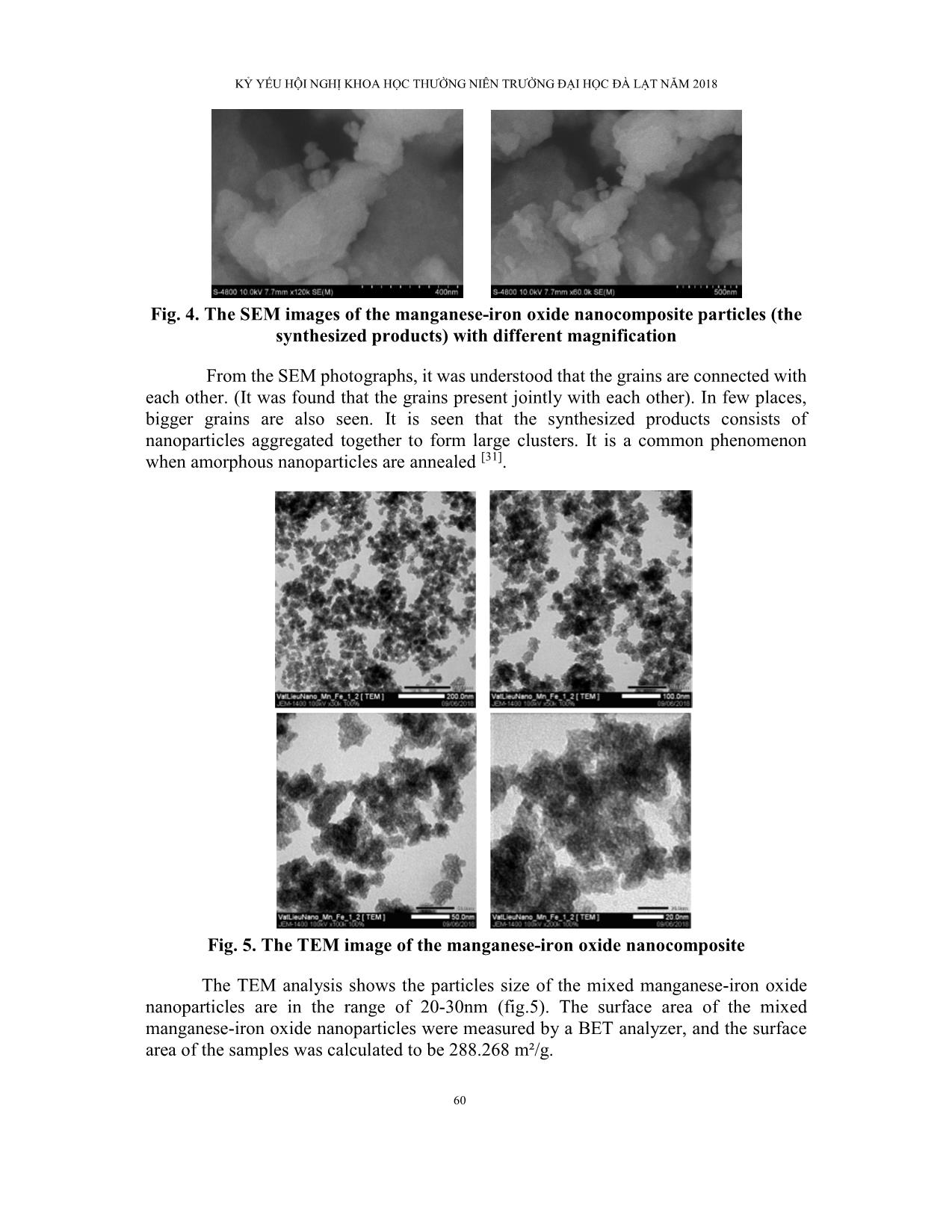
Trang 8
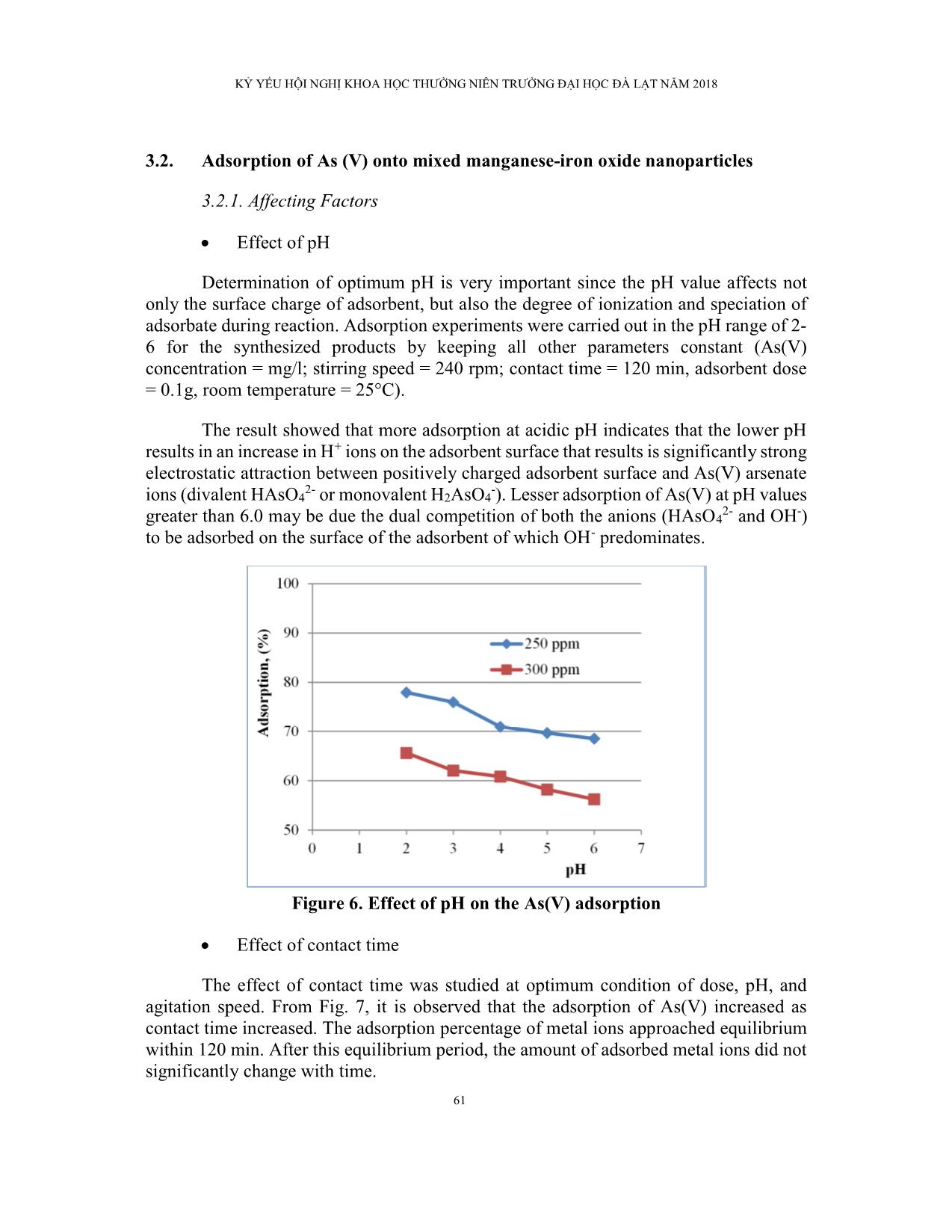
Trang 9
Trang 10
Tải về để xem bản đầy đủ
Tóm tắt nội dung tài liệu: Synthesis and application of mixed manganese - Iron oxide nanoparticles for adsorption of as(v) from aqueous solutions

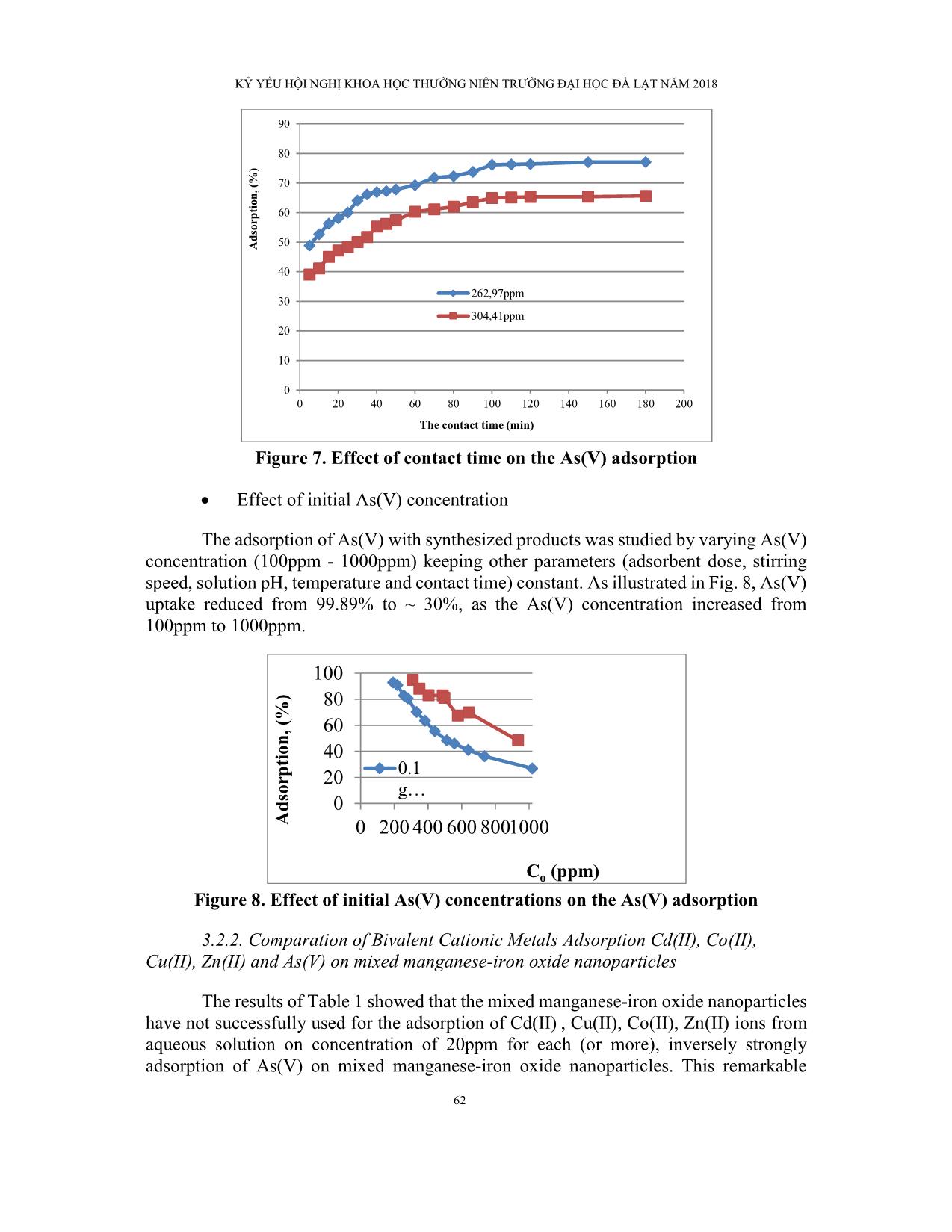
KỶ YẾU HỘI NGHỊ KHOA HỌC THƯỜNG NIÊN TRƯỜNG ĐẠI HỌC ĐÀ LẠT NĂM 2018 53 SYNTHESIS AND APPLICATION OF MIXED MANGANESE-IRON OXIDE NANOPARTICLES FOR ADSORPTION OF As(V) FROM AQUEOUS SOLUTIONS Synthesis of adsorbent and its adsorption Le Ngoc Chunga*, Le Thanh Quoca aThe Faculty of Chemistry, Dalat University, Lamdong, Vietnam Correspoding author: Email: [email protected] Abstract A simple method has been used to synthesize nanoparticles of mixed manganese-iron oxide for the adsorption of As(V) metal ions from aqueous solutions. Transmission Electron Microscopy (TEM), X-Ray diffraction (XRD), Scanning Electron Microscopy (SEM), Fourier transform infrared spectroscopy (FTIR), BET analysis were used to determine particle size and characterization of produced nanoparticles. The x-ray diffraction pattern indicated that the as-synthesized adsorbent is amorphous with 288.268 m2/g surface area; the amorphous synthesized products were aggregated with many nanosized particles. The crystallinity of the Mn2O3/Fe2O3 were obtained at 400°C and 600°C calcination temperature. The FTIR spectra confirmed the presence of -OH group and H-O-H group localized at 3200 - 3400 cm–1 and 1618-1653 cm−1; theses intense bands is weak (fade) at the high calcination temperature of the mixed manganese-iron oxide nanoparticles. In addition, when the calcination temperature of the mixed manganese-iron oxide nanoparticles was 400OC, the weak absorption bands at 630 cm−1 due to the vibrations of (Fe-O). The results showed that the mixed manganese-iron oxide nanoparticles has high selectivity for As(V). Keywords: Mixed manganese-iron oxide nanoparticles; Amorphous; As(V). KỶ YẾU HỘI NGHỊ KHOA HỌC THƯỜNG NIÊN TRƯỜNG ĐẠI HỌC ĐÀ LẠT NĂM 2018 54 TỔNG HỢP VÀ ỨNG DỤNG HỖN HỢP NANO OXID MANGAN- SẮT ĐỂ HẤP PHỤ As(V) TỪ DUNG DỊCH NƯỚC Tổng hợp chất hấp phụ và tính chất hấp phụ Lê Ngọc Chunga*, Lê Thành Quốca aKhoa Hóa học, Trường Đại học Đà Lạt, Lâm Đồng, Việt Nam *Tác giả liên hệ: Email: [email protected] Tóm tắt Một phương pháp đơn giản cho sự tổng hợp chất hấp phụ hỗn hợp nano oxid mangan-sắt để hấp phụ ion kim loại As(V) từ dung dịch nước. Phương pháp kính hiển vi điện tử truyền qua (TEM), nhiễu xạ tia X (XRD), kính hiển vi điện tử quét (SEM), phổ hồng ngoại (FTIR), phân tích BET được sử dụng để xác định kích thước hạt và đặc trưng của hỗn hợp nano oxid mangan-sắt. Nhiễu xạ tia X cho thấy chất hấp phụ tổng hợp là hỗn hợp nano oxid mangan- sắt có cấu trúc vô định định hình có diện tích bề mặt 288.268 m2/g và bị hiện tượng aggregation. Khi nung hỗn hợp nano oxid mangan-sắt ở nhiệt độ 400OC và 600OC sẽ xuất hiện cấu trúc của tinh thể Mn2O3/Fe2O3. Phổ hồng ngoại FTIR cũng xác nhận hỗn hợp nano oxid mangan-sắt được tổng hợp có sự hiện diện của nhóm –OH và nhóm H-O-H tại dải hấp thụ 3200 - 3400 cm–1 và 1618-1653 cm−1; cường độ của dải hấp thụ này sẽ yếu đi khi hỗn hợp nano oxid mangan-sắt nung ở nhiệt độ cao. Hơn nữa, khi hỗn hợp nano oxid mangan- sắt nung đến nhiệt độ 400OC thì xuất hiện peak hấp thụ yếu tại 630 cm−1 gây nên do nhóm Fe-O. Kết quả cũng chỉ rằng hỗn hợp nano oxid mangan-sắt có tính chọn lọc cao đối với As(V). Keywords: Mixed manganese-iron oxide nanoparticles, Amorphous , As(V). KỶ YẾU HỘI NGHỊ KHOA HỌC THƯỜNG NIÊN TRƯỜNG ĐẠI HỌC ĐÀ LẠT NĂM 2018 55 1. INTRODUCTION Water plays important roles in the natural environment, human activities, and social development. However, the presence of arsenic in natural waters has become a worldwide problem in the past decades [1,2]. Arsenic pollution has been reported recently in USA, China, Chile, Bangladesh, Taiwan, Mexico, Romania, United Kingdom, Argentina, Poland, Canada, Hungary, New Zealand, Vietnam, Cambodia, Japan and India [1-7]. Arsenic commonly exists as two inorganic forms of arsenite (AsO33−) and arsenate (AsO43−) which are the popular forms in water and referred to as As(III) and As(V). In general, As(V) is stable in aerobic environment and As(III) often exists in anaerobic environment. The toxicity of arsenic species is different, generally the toxicity of inorganic arsenic compounds is about 100 times higher than organic arsenic compounds, and the toxicity of inorganic As(III) compounds are approximately 60–80 times higher to humans than As(V) compounds [4-10]. They causes skin, lung, bladder and kidney cancer as well as pigmentation changes, skin thickening (hyperkeratosis), neurological disorders, muscular weakness, loss of appetite and nausea [5-12]. Therefore, it is really necessary to remove arsenic from water to make sure that our environment is safe. Adsorption has been recognized as a promising technique for removing arsenic from drinking water due to its high removal capacity and ease of operation. However, As(III) is less efficiently removed than As(V) from aqueous solutions by almost all of the arsenic removal technologies and pre-oxidation of As(III) to As(V) is required [4-17]. Recently, increasing attention has been focused on metal oxide sorbents such as iron, aluminum, titanium, manganese, and zirconium. Among these iron oxides were the mostly studied because of their high affinity to arsenic species, low cost and environmental friendliness [8-21]. Most recently, many researchers used metal composite materials (containing two or more metals) as adsorbents to remove As from contaminated water. The results showed that the composite metal oxides can not only inherit the advantages of parent oxides but also show a synergistic effect of higher adsorption capacity than that of individual metal oxides (Lata and Samadder 2016). For instances, Zhang et al. (2005) developed an Fe-Ce bimetal oxide sorbent, which has a much higher As(V) adsorption capacity than the individual Ce and Fe oxide. Zhang et al. (2007) prepared an Fe-Mn binary oxide sorbent, exhibiting a greater enhancement in both As(V) and As(I ... posite particles (the synthesized products) with different magnification From the SEM photographs, it was understood that the grains are connected with each other. (It was found that the grains present jointly with each other). In few places, bigger grains are also seen. It is seen that the synthesized products consists of nanoparticles aggregated together to form large clusters. It is a common phenomenon when amorphous nanoparticles are annealed [31]. Fig. 5. The TEM image of the manganese-iron oxide nanocomposite The TEM analysis shows the particles size of the mixed manganese-iron oxide nanoparticles are in the range of 20-30nm (fig.5). The surface area of the mixed manganese-iron oxide nanoparticles were measured by a BET analyzer, and the surface area of the samples was calculated to be 288.268 m²/g. KỶ YẾU HỘI NGHỊ KHOA HỌC THƯỜNG NIÊN TRƯỜNG ĐẠI HỌC ĐÀ LẠT NĂM 2018 61 3.2. Adsorption of As (V) onto mixed manganese-iron oxide nanoparticles 3.2.1. Affecting Factors Effect of pH Determination of optimum pH is very important since the pH value affects not only the surface charge of adsorbent, but also the degree of ionization and speciation of adsorbate during reaction. Adsorption experiments were carried out in the pH range of 2- 6 for the synthesized products by keeping all other parameters constant (As(V) concentration = mg/l; stirring speed = 240 rpm; contact time = 120 min, adsorbent dose = 0.1g, room temperature = 25°C). The result showed that more adsorption at acidic pH indicates that the lower pH results in an increase in H+ ions on the adsorbent surface that results is significantly strong electrostatic attraction between positively charged adsorbent surface and As(V) arsenate ions (divalent HAsO42- or monovalent H2AsO4-). Lesser adsorption of As(V) at pH values greater than 6.0 may be due the dual competition of both the anions (HAsO42- and OH-) to be adsorbed on the surface of the adsorbent of which OH- predominates. Figure 6. Effect of pH on the As(V) adsorption Effect of contact time The effect of contact time was studied at optimum condition of dose, pH, and agitation speed. From Fig. 7, it is observed that the adsorption of As(V) increased as contact time increased. The adsorption percentage of metal ions approached equilibrium within 120 min. After this equilibrium period, the amount of adsorbed metal ions did not significantly change with time. 50 60 70 80 90 100 0 1 2 3 4 5 6 7 A ds or pt io n, (% ) pH 250 ppm 300 ppm KỶ YẾU HỘI NGHỊ KHOA HỌC THƯỜNG NIÊN TRƯỜNG ĐẠI HỌC ĐÀ LẠT NĂM 2018 62 Figure 7. Effect of contact time on the As(V) adsorption Effect of initial As(V) concentration The adsorption of As(V) with synthesized products was studied by varying As(V) concentration (100ppm - 1000ppm) keeping other parameters (adsorbent dose, stirring speed, solution pH, temperature and contact time) constant. As illustrated in Fig. 8, As(V) uptake reduced from 99.89% to ~ 30%, as the As(V) concentration increased from 100ppm to 1000ppm. Figure 8. Effect of initial As(V) concentrations on the As(V) adsorption 3.2.2. Comparation of Bivalent Cationic Metals Adsorption Cd(II), Co(II), Cu(II), Zn(II) and As(V) on mixed manganese-iron oxide nanoparticles The results of Table 1 showed that the mixed manganese-iron oxide nanoparticles have not successfully used for the adsorption of Cd(II) , Cu(II), Co(II), Zn(II) ions from aqueous solution on concentration of 20ppm for each (or more), inversely strongly adsorption of As(V) on mixed manganese-iron oxide nanoparticles. This remarkable 0 10 20 30 40 50 60 70 80 90 0 20 40 60 80 100 120 140 160 180 200 A ds or pt io n, (% ) The contact time (min) 262,97ppm 304,41ppm 0 20 40 60 80 100 0 200 400 600 8001000A ds or pt io n, (% ) Co (ppm) 0.1 g KỶ YẾU HỘI NGHỊ KHOA HỌC THƯỜNG NIÊN TRƯỜNG ĐẠI HỌC ĐÀ LẠT NĂM 2018 63 difference is probably due to the difference ionic radius (table 2) and the greater the valence. Danny et al. (2004) and Lee and Moon (2001) explained that the smaller the ionic radius and the greater the valence, the more closely and strongly is the ion adsorbed [32-34]. Therefore, the mixed manganese-iron oxide nanoparticles have successfully used for the adsorption of As(V) from aqueous solution. Table 1. Adsorption percentage of metal ions on to mixed manganese-iron oxide nanoparticles pH Adsorption of metal ions on to mixed manganese-iron oxide nanoparticles, (%) As(V) (150ppm) Cd(II) (20ppm) Co(II) (20ppm) Cu(II) (20ppm) Zn(II) (20ppm) Adsorption percentage, % SD* 2 99.89 0.01 non-ads. non-ads. - - 3 98.45 0.09 non-ads. non-ads. - - 4 95.28 0.05 non-ads. non-ads. non-ads. non-ads. 5 96.46 0.06 non-ads. non-ads. - - 6 96.51 0.01 non-ads. non-ads. - - Note: (*) Standard deviation (SD) Table 2. Effective ionic radii in pm of elements Ion As(V) Cd(II) Co(II) Cu(II) Zn(II) Effective ionic radii (pm) 46 95 65 73 74 4. CONCLUSION A simple method has been used to synthesize nanoparticles of mixed manganese- iron oxide for the adsorption of As(V) metal ions from aqueous solutions under batch conditions. Transmission Electron Microscopy (TEM), X-Ray diffraction (XRD), Scanning Electron Microscopy (SEM), Fourier transform infrared spectroscopy (FTIR), BET analysis were used to determine particle size and characterization of produced nanoparticles. Moreover, the effects of pH, contact time and adsorbent weight on adsorption process were investigated. The high uptake of As(V) by the mixed manganese- iron oxide nanoparticles may be due to its relatively high surface area. REFERENCES 1- Pankaj Verma, Abhinay Agarwal and V.K.Singh (2014) Arsenic removal from water through adsorption-A Review. Recent Research in Science and Technology. 6(1), pp. 219-226. KỶ YẾU HỘI NGHỊ KHOA HỌC THƯỜNG NIÊN TRƯỜNG ĐẠI HỌC ĐÀ LẠT NĂM 2018 64 2- Dinesh Mohan, Charles U. Pittman Jr. (2007). Arsenic removal from water/wastewater using adsorbents-A critical review. Journal of Hazardous Materials. 142, pp. 1–53. 3- Saikia Ananta, Banerjee Saumen and Veer Vijay. (2015). Adsorption Isotherm, Thermodynamic and Kinetic Study of Arsenic (III) on Iron Oxide Coated Granular Activated Charcoal. International Research Journal of Environment Sciences. Vol. 4(1), pp. 64-77. 4- Yumei Kang. (2016) Arsenic-Polluted Groundwater in Cambodia: Advances in Research. International Journal of Water and Wastewater Treatment. Review Article. Vol. 2(1): doi org/10.16966/2381-5299.116 Open Access 2. 5- Sanjoy Kumar Maji, Anjali Pal, Tarasankar Pal, Asok Adak. (2007). Adsorption thermodynamics of arsenic on Laterite soil. J. Surface Sci. Technol. Vol. 22, No.3- 4, pp. 161-176. 6- Branislava M. Jovanović, Vesna L. Vukašinović-Pešić, Đorđe N. Veljović, Ljubinka V. Rajaković. (2011) Arsenic removal from water using low-cost adsorbents – A comparative study. J. Serb. Chem. Soc. 76 (10), pp. 1437–1452. 7- Andjelkovic L., Manojlovic D., Skrivanj S., Pavlovic B. M., Amaizah N. R., Roglic G. (2013) As(III) and As(V) Sorption on MnO2 Synthesized by Mechano- chemical Reaction from Aqueous phase. Int. J. Environ. Res. 7(2), pp. 395-402. 8- Shugi Kuriakose, Tony Sarvinder Singh and Kamal K. Pant. (2004) Adsorption of As(III) from Aqueous Solution onto Iron Oxide Impregnated Activated Alumina. Water Qual. Res. J. Canada. Volume 39, No. 3, pp. 258–266. 9- Can Wang, Hanjin Luoa, Zilong Zhang, Yan Wua, Jian Zhang, Shaowei Chena. (2014) Removal of As(III) and As(V) from aqueous solutions using nanoscalezero valent iron-reduced graphite oxide modified composites. Journal of Hazardous Materials. 268, pp. 124– 131. 10- Suyun He, Caiyun Han, Sufang He, Hua Wang, Chunxia Liu,Yun Shan, Yongming Luo. (2013) Kinetic study of arsenic(V) absorption with Al2O3 functionalized SBA-15. Advanced Materials Research. Vols. 726-731, pp. 2191- 2197. 11- Younghun Kim, Changmook Kim, Inhee Choi, Selvaraj Rengaraj, Jongheop Yi. (2004) Arsenic Removal Using Mesoporous Alumina Prepared via a Templating Method. Environ. Sci. Technol. 38, pp. 924-931. 12- Shuqiong Kong, Yanxin Wang, Hongbin Zhan, Songhu Yuan, Mei Yu, Mingliang Liu. (2014) Adsorption/Oxidation of Arsenic in Groundwater by Nanoscale Fe- Mn Binary Oxides Loaded on Zeolite. Water Environment Research. Volume 86, Number 2, pp.147 – 155. 13- Qingxiang Zhou, Jingjie Wang, Xiangkun Liao, Junping Xiao, Huili Fan. (2015) Removal of As(III) and As(V) from Water Using Magnetic Core-Shell KỶ YẾU HỘI NGHỊ KHOA HỌC THƯỜNG NIÊN TRƯỜNG ĐẠI HỌC ĐÀ LẠT NĂM 2018 65 Nanomaterial Fe3O4@Polyaniline. International Journal of Green Technology.Vol. 1, pp. 54-64. 14- Daryoush Afzalia, Mina Rouhanic, Fariba Fathiradc, Tayebeh Shamspurc, Ali Mostafavic. (2016) Nano-iron oxide coated on sand as a new sorbent for removal of arsenic from drinking water. Desalination and Water Treatment. 57, pp. 13030–13037. 15- Aamir D. Abid, Masakazu Kanematsu, Thomas M. Young, Ian M. Kennedy. (2013) Arsenic removal from water using flame-synthesized iron oxide nanoparticles with variable oxidation states. Aerosol Sci Technol. 47(2), pp. 169– 176. 16- Douglas Oti, Joniqua Howard, Erlande Omisca Ken Thomas and Maya Trotz. (2011) Adsorption of Arsenite onto Kemiron. African Journal of Environmental Science and Technology. Vol. 5(12), pp. 1060-1068. 17- A. R. Rahmani, H.R. Ghaffari, M.T. Samadi. A comparative study on arsenic (III) removal from aqueous solution using nano and micro sized zero- valent iron. (2011) Iran. J. Environ. Health. Sci. Eng. Vol. 8, No. 2, pp. 175-180. 18- Lei Chen, Hongchuan Xin, Yuan Fang, Cong Zhang, Feng Zhang, Xing Cao, Chunhui Zhang, Xuebing Li. (2014) Application of Metal Oxide Heterostructures in Arsenic Removal from Contaminated Water. Journal of Nanomaterials. Review Article. Volume 2014, Article ID 793610, 10 pages. 19- Sang-Yoon Min, Byeong-Kwon Kim, Sun-Ju Park, Yoon-Young Chang, Jae Kyu Yang. (2009) Removal Efficiency of Arsenic by Adsorbents having Different Type of Metal Oxides. Environ. Eng. Res. Vol. 14, No. 2, pp. 134-139. 20- Sneh Lata, S.R. Samadder. (2016) Removal of arsenic from water using nano adsorbents and challenges: A review. Journal of Environmental Management. Vol.166, pp. 387-406. 21- Daniel Ocinski, Irena Jacukowicz-Sobala, Piotr Mazur, Jerzy Raczyk, Elzbieta Kociołek-Balawejder. (2016) Water treatment residuals containing iron and manganese oxides for arsenic removal from water – Characterization of physicochemical properties and adsorption studies. Chemical Engineering Journal. Vol.294, pp. 210–221. 22- Wensheng Zhang, Pritam Singh, Touma B. Issa. (2011) Arsenic(III) Remediation from Contaminated Water by Oxidation and Fe/Al Co-Precipitation. Journal of Water Resource and Protection. Vol. 3, 655-660. 23- Gaosheng Zhang, Huijuan Liu, Ruiping Liu, Jiuhui Qu. (2009) Adsorption behavior and mechanism of arsenate at Fe–Mn binary oxide/water interface. Journal of Hazardous Materials. Vol.168, pp. 820–825. 24- Gaosheng Zhang, Huijuan Liu, Jiuhui Qu, William Jefferson. (2012) Arsenate uptake and arsenite simultaneous sorption and oxidation by Fe–Mn binary oxides: KỶ YẾU HỘI NGHỊ KHOA HỌC THƯỜNG NIÊN TRƯỜNG ĐẠI HỌC ĐÀ LẠT NĂM 2018 66 Influence of Mn/Fe ratio, pH, Ca2+, and humic acid. Journal of Colloid and Interface Science. 366, pp. 141–146. 25- Gaosheng Zhang, Zongming Ren, Xiwang Zhang, Jing Chen. (2013) Nanostructured iron(III)-copper(II) binary oxide: A novel adsorbent for enhanced arsenic removal from aqueous solutions. Water re search. Vol. 47, pp. 4022- 4031. 26- Shuqiong Kong, YanxinWang, Hongbin Zhana, Mingliang Liu, Lili Liang, Qinhong Hu. (2014) Competitive adsorption of humic acid and arsenate on nanoscale iron–manganese binary oxide-loaded zeolite in groundwater. Journal of Geochemical Exploration. 144, pp. 220–225. 27- Ngoc Chung Le and Dinh Van Phuc. (2015) Sorption of Lead (II), Cobalt (II) and Copper (II) ions from aqueous solutions by nanostructured gamma – MnO2. Advances in Natural Sciences: Nanoscience and Nanotechnology, issue 2, 6, 025014 (8pp), ISSN: 2043-6262, doi:10.1088/2043-6262/6/2/025014. 28- Van-Phuc Dinh, Ngoc-Chung Le, Thi-Phuong-Tu Nguyen, Thi-Dong-Thuong Hoang, Van-Dong Nguyen, Ngoc-Tuan Nguyen. (2016) Zinc Adsorption Property of Gamma-MnO2 Nanostructure: Equilibrium and Kinetic Studies. Key Engineering Materials. Vol. 708, pp 3-8. 29- Van-Phuc Dinh, Ngoc-Chung Le, Thi-Phuong-Tu Nguyen, Ngoc-Tuan Nguyen. (2016) Synthesis of 𝛼-MnO2 Nanomaterial from a Precursor 𝛾-MnO2: Characterization and Comparative Adsorption of Pb(II) and Fe(III). Journal of Chemistry. Vol. 2016, Article ID 8285717, 9 pages. 30- Haile Hasana Logita, Abi Tadesse,Tesfahun Keb. (2015) Synthesis, characterization and photocatalytic activity of MnO2/Al2O3/Fe2O3 nanocomposite for degradation of malachite green. African Journal of Pure and Applied Chemistry. Vol. 9(11), pp. 211-222. 31- Jriuan Lai, Kurikka V. P. M. Shafi, Abraham Ulman, Katja Loos, Nan-Loh Yang, Min-Hui Cui, Thomas Vogt, Claude Estourne`s, Dave C. Locke. (2004) Mixed Iron-Manganese Oxide Nanoparticles. J. Phys. Chem. B. Vol. 108, pp. 14876- 14883. 32- Danny, C. K., Chun, K., Cheung, W., Keith, K. H., John, F. and McKay, G. (2004) Sorption equilibria of metal ions on bone. char. Chemosphere. Vol. 54, pp. 273– 281. 33- Dae Haeng Lee, Hee Moon. (2001) Adsorption Equilibrium of Heavy Metals on Natural Zeolites. Korean J. Chem. Eng., 18(2), 247-256. 34- S. B. Chen, Y. B. Ma, L. Chen, K. Xian. (2010) Adsorption of aqueous Cd2+, Pb2+, Cu2+ ions by nano-hydroxyapatite: Single- and multi-metal competitive adsorption study. Geochemical Journal, Vol. 44, pp. 233-239.
File đính kèm:
 synthesis_and_application_of_mixed_manganese_iron_oxide_nano.pdf
synthesis_and_application_of_mixed_manganese_iron_oxide_nano.pdf

