A survey on some immune indicators in the group of vietnamese children who did not respond to rotavac vaccine
To investigate the expression of beta 7 integrin on B and T lymphocytes using a flow cytometry system. Subjects and methods: We analyzed immunological parameters such as B and T lymphocyte population as well as B and T cell subpopulations expressing beta 7 integrin molecules by flow cytometry on the two groups of children with and without response to the Rotavirus vaccine. Results: The number of peripheral blood lymphocytes of the non-Response group was statistically significantly lower than that of the response group ( ± SD: 4.89 ± 1.71 vs 5.77 ± 1.46 million/mL, p = 0.04). The number of T cells (CD3+) of the non-response group was statistically significantly lower than that of the response group ( ± SD: 1.65 ± 0.61 vs 2.05 ± 0.64 million/mL, p = 0.018). The frequency and number of T-cells expressing beta 7 integrin of the non-response group were statistically significantly lower than those of the response group (Frequency ( ± SD): 17.48 ± 2.89 vs 19.44 ± 3.74%, p = 0.029; Quantity ( ± SD): 0.87 ± 0.41 vs 1.13 ± 0.37 million/mL, p = 0.014). There was no statistically significant difference between 2 groups for B-cell-related parameters in peripheral blood such as the number and frequency of total B cells and the B-cell subpopulation expressing beta 7 integrin in peripheral blood. Conclusion: The group of children not responding to the Rotavirus vaccine was statistically significantly lower than that of the responding group for the following parameters: Total lymphocyte count; frequency and number of the subpopulation of T-lymphocytes expressing 7 beta integrin in peripheral blood

Trang 1

Trang 2
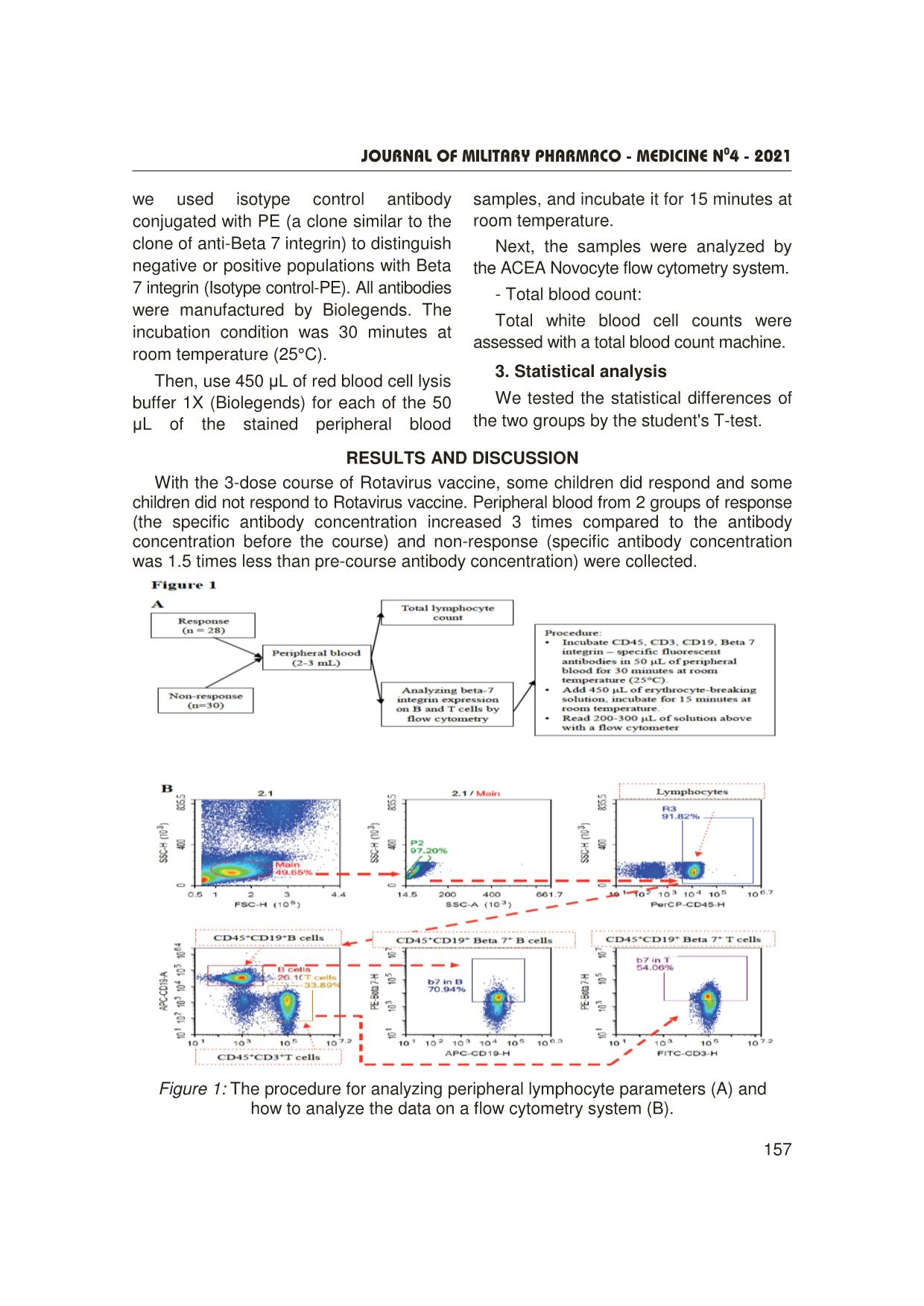
Trang 3
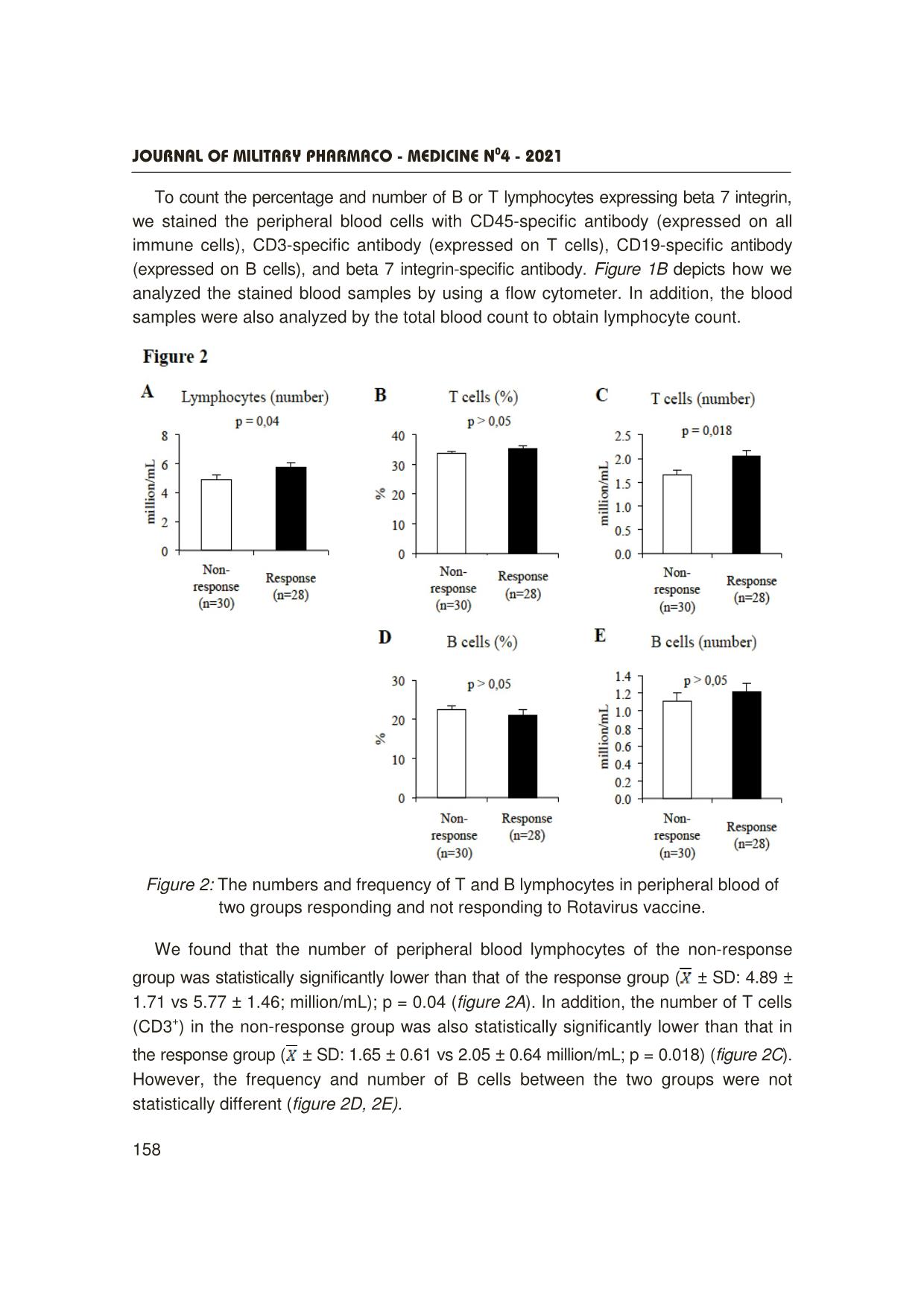
Trang 4
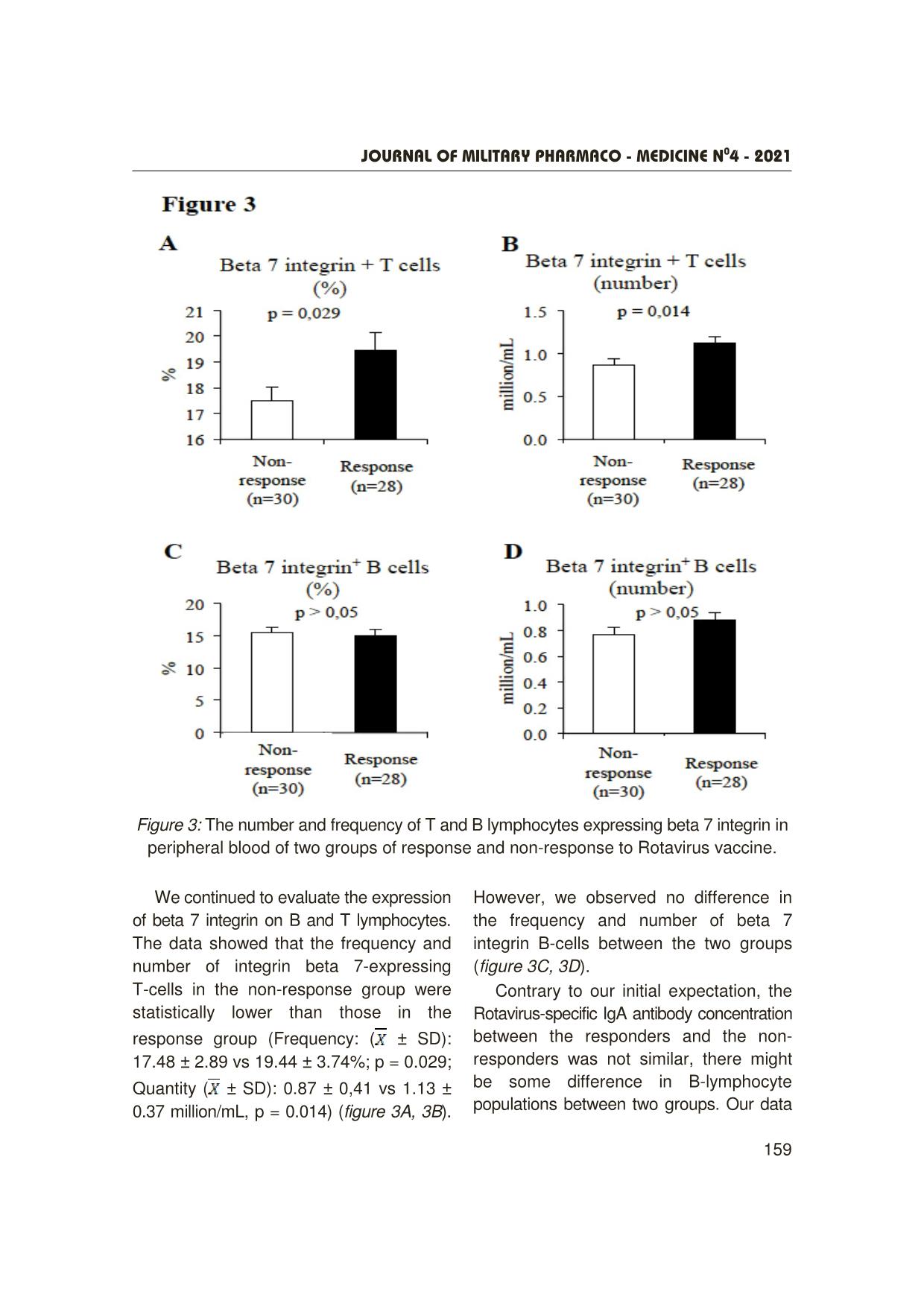
Trang 5

Trang 6

Trang 7
Tóm tắt nội dung tài liệu: A survey on some immune indicators in the group of vietnamese children who did not respond to rotavac vaccine

Journal OF MILITARY PHARMACO - MEDICINE N04 - 2021 155 A SURVEY ON SOME IMMUNE INDICATORS IN THE GROUP OF VIETNAMESE CHILDREN WHO DID NOT RESPOND TO ROTAVAC VACCINE Nguyen Minh Hai1, Phung The Hai³, Do Khac Dai³ Pham Minh Dam1, Pham Ngoc Hung², Nguyen Dang Dung³ SUMMARY Objectives: To investigate the expression of beta 7 integrin on B and T lymphocytes using a flow cytometry system. Subjects and methods: We analyzed immunological parameters such as B and T lymphocyte population as well as B and T cell subpopulations expressing beta 7 integrin molecules by flow cytometry on the two groups of children with and without response to the Rotavirus vaccine. Results: The number of peripheral blood lymphocytes of the non-response group was statistically significantly lower than that of the response group ( ± SD: 4.89 ± 1.71 vs 5.77 ± 1.46 million/mL, p = 0.04). The number of T cells (CD3+) of the non-response group was statistically significantly lower than that of the response group ( ± SD: 1.65 ± 0.61 vs 2.05 ± 0.64 million/mL, p = 0.018). The frequency and number of T-cells expressing beta 7 integrin of the non-response group were statistically significantly lower than those of the response group (Frequency ( ± SD): 17.48 ± 2.89 vs 19.44 ± 3.74%, p = 0.029; Quantity ( ± SD): 0.87 ± 0.41 vs 1.13 ± 0.37 million/mL, p = 0.014). There was no statistically significant difference between 2 groups for B-cell-related parameters in peripheral blood such as the number and frequency of total B cells and the B-cell subpopulation expressing beta 7 integrin in peripheral blood. Conclusion: The group of children not responding to the Rotavirus vaccine was statistically significantly lower than that of the responding group for the following parameters: Total lymphocyte count; frequency and number of the subpopulation of T-lymphocytes expressing 7 beta integrin in peripheral blood. * Keywords: Rotavirus vaccine; Beta 7 integrin; β7- integrin; B lymphocytes; T lymphocytes. INTRODUCTION Rotavirus is a common cause of diarrhea among children globally as well as in Vietnam [1]. Currrently, orally live- attenuated Rotavirus vaccines have been proven effective and widely used [2]. However, there is a proportion of non- responders to these vaccines, particularly in low-income countries such as in Africa and Asia, where the efficacy ranges from 51% to 64% on average [3, 4, 5]. Factors influencing vaccine efficacy may include: immune status of the body, general nutritional status, nature of circulating rotavirus strains, intestinal microbiota, etc. 1Department of Exams Accreditation and Medical Education Quality Assurance, Vietnam Military Medical University 2Department of Epidemiology, Vietnam Military Medical University ³Department of Immunology, Vietnam Military Medical University Corresponding author: Nguyen Minh Hai ([email protected]) Date received: 03/02/2021 Date accepted: 10/4/2021 Journal OF MILITARY PHARMACO - MEDICINE N04 - 2021 156 Based on a clinical trial study of ROTAVAC vaccine in children aged 6 - 8 weeks, we conducted this study: To investigate a number of factors affecting the immunogenicity of the rotavirus vaccine in Vietnamese children. After oral vaccination of Rotavirus vaccine, viral antigens were captured and presented by antigen-presenting cells (APCs). The APCs then migrate to the intestinal secondary lymphoid organs or mesenteric lymph nodes to present viral antigens to specific B and T cells, resulting in activation of the humoral immune response (antibody production) and the cell-mediated immune response (helper T and cytotoxic T cells) specific to Rotavirus. The specific B and T cells then differentiate, proliferate, and move into the blood, before moving through the intestinal capillaries and retaining in the intestine. The gut-homing process of specific B and T lymphocytes depends on the expression of an important molecule, beta 7 integrin. Beta 7 integrin can combine with another molecule, alpha 4 integrin on lymphocytes to specifically bind to MAdCAM-1 molecule (expressed only on intestinal capillary endothelial cells). After penetrating the intestinal capillaries, beta 7 integrin can also combine with alpha E integrin molecule to specifically bind E-cadherin molecule (expressed only on intestinal epithelial cells), by which lymphocytes B or T can be anchored in the intestine to perform their task [6]. We hypothesized that there might be a difference in the expression of beta 7 integrin molecule on the surface of B and T lymphocytes (which plays an important role in the humoral immune response) between the two groups of children with response and non-response to Rotavirus vaccine. Therefore, this study was carried out to investigate the expression of beta 7 integrin on B and T lymphocytes using a flow cytometry system. SUBJECTS AND METHODS 1. Subject, time, place of the study - Study subjects: 58 children receiving ROTAVAC® vaccine and having been tested for IgA with the results of antibody titration after vaccination who were divided into two groups: Group 1 (n = 30): With very low initial antibody titres and/or the ratio after/before vaccination below 1.5. Group 2 (n = 28): With the relatively high initial antibody titres and/or the ratio after/before vaccination ≥ 3 [7]. - Location: The study was conducted in Hung Ha district, Thai Binh province. - Time: From June 2020 to July 2020. After using ROTAVAC® vaccine (3 doses enough), children were given 2 - 3 mL of EDTA blood to analyze some parameters of immune cells by flow cytometry or total blood count. Collected samples were coded and double-blindly analyzed. 2. Methods - A protocol of evaluating lymphocytes with a flow cytometry system: 50 µL of peripheral blood of each study subject was incubated with fluorescent antibodies (Abs) including anti-CD45 antibody with PerCP (anti-CD45-PerCP), anti-CD3 antibody with FITC (anti-CD3- FITC), anti-CD19 antibody with APC (anti-CD19-APC) and anti-beta 7 integrin with PE (anti-beta 7 integrin-PE). In addition, we used isotype control antibody conjugated with PE (a clone similar to the clone of anti-beta 7 integrin) to distinguish negative or positive populations with Beta Journal OF MILITARY PHARMACO - MEDICINE N04 - 2021 157 we used isotype control antibody conjugated with PE (a clone similar to the clone of anti-Beta 7 integrin) to distinguish negative or positive populations with Beta 7 integrin (Isotype control-PE). All antibodies were manufactured by Biolegends. The incubation condition was 30 minutes at room temperature (25°C). Then, use 450 µL of red blood cell lysis buffer 1X (Biolegends) for each of the 50 µL of the stained peripheral blood samples, and incubate it for 15 minutes at room temperature. Next, the samples were analyzed by the ACEA Novocyte flow cytometry system. - Total blood count: Total white blood cell counts were assessed with a total blood count machine. 3. Statistical analysis We tested the statistical differences of the two groups by the student's T-test. RESULTS AND DISCUSSION With the 3-dose course of Rotavirus vaccine, some children did respond and some children did not respond to Rotavirus vaccine. Peripheral blood from 2 groups of response (the specific antibody concentration increased 3 times compared to the antibody concentration before the course) and non-response (specific antibody concentration was 1.5 times less than pre-course antibody concentration) were collected. Figure 1: The procedure for analyzing peripheral lymphocyte parameters (A) and how to analyze the data on a flow cytometry system (B). Journal OF MILITARY PHARMACO - MEDICINE N04 - 2021 158 To count the percentage and number of B or T lymphocytes expressing beta 7 integrin, we stained the peripheral blood cells with CD45-specific antibody (expressed on all immune cells), CD3-specific antibody (expressed on T cells), CD19-specific antibody (expressed on B cells), and beta 7 integrin-specific antibody. Figure 1B depicts how we analyzed the stained blood samples by using a flow cytometer. In addition, the blood samples were also analyzed by the total blood count to obtain lymphocyte count. Figure 2: The numbers and frequency of T and B lymphocytes in peripheral blood of two groups responding and not responding to Rotavirus vaccine. We found that the number of peripheral blood lymphocytes of the non-response group was statistically significantly lower than that of the response group ( ± SD: 4.89 ± 1.71 vs 5.77 ± 1.46; million/mL); p = 0.04 (figure 2A). In addition, the number of T cells (CD3+) in the non-response group was also statistically significantly lower than that in the response group ( ± SD: 1.65 ± 0.61 vs 2.05 ± 0.64 million/mL; p = 0.018) (figure 2C). However, the frequency and number of B cells between the two groups were not statistically different (figure 2D, 2E). Journal OF MILITARY PHARMACO - MEDICINE N04 - 2021 159 Figure 3: The number and frequency of T and B lymphocytes expressing beta 7 integrin in peripheral blood of two groups of response and non-response to Rotavirus vaccine. We continued to evaluate the expression of beta 7 integrin on B and T lymphocytes. The data showed that the frequency and number of integrin beta 7-expressing T-cells in the non-response group were statistically lower than those in the response group (Frequency: ( ± SD): 17.48 ± 2.89 vs 19.44 ± 3.74%; p = 0.029; Quantity ( ± SD): 0.87 ± 0,41 vs 1.13 ± 0.37 million/mL, p = 0.014) (figure 3A, 3B). However, we observed no difference in the frequency and number of beta 7 integrin B-cells between the two groups (figure 3C, 3D). Contrary to our initial expectation, the Rotavirus-specific IgA antibody concentration between the responders and the non- responders was not similar, there might be some difference in B-lymphocyte populations between two groups. Our data Journal OF MILITARY PHARMACO - MEDICINE N04 - 2021 160 showed that B cells between the two groups showed no difference in the frequency and number of total B cells as well as the beta 7 integrin expressing B-cell subpopulation (figures 2D, 2E, 3C, 3D). Interestingly, we observed a difference in the beta 7 integrin expressing T subpopulation, in which the non-response group showed lower frequency and number than the response group. (figures 3A, 3B). Although the role of secreting antibodies specific to Rotavirus (IgA, IgG) is predominant due to the specific B cells, the specific T cells may also play a role in assisting B cell proliferation and differentiation. This phenomenon occurs in the secondary lymphoid organs in the intestine such as the Payer patches or the mesenteric lymph nodes. The difficulties in accessing to these human tissues hinder understanding of the specific molecular biology mechanisms behind the vaccine unresponsiveness. Our results suggest further studies on the correlation between expression of the beta 7 integrin molecule on T cells and the specific antibody immune response. On the other hand, many studies have shown the important role of the vitamin A/rentinoic acid axis (a product of vitamin A metabolism via an enzyme retinal dehydrogenase in antigen- presenting cells) in the expression of molecules that are important for T lymphocytes to return to the intestine such as beta 7 integrin or CCR9 on T cells, as well as MAd-CAM-1 on intestinal vascular endothelial cells, and CCL25 (bound to CCR9) by the intestinal mucosa secreted [8, 9]. Our study suggests that vitamin A deficiency in young children should be further explored when using oral vaccines absorbed through the intestinal mucosa. CONCLUSION The group of children who did not respond to the Rotavirus vaccine showed statistically significantly lower parameters compared to the response group including total lymphocyte count; frequency and number of subpopulation of T-lymphocytes expressing an 7-beta integrin in peripheral blood. There was no statistically significant difference between the two groups for peripheral blood B-cell-related parameters such as the number and frequency of total B cells and the B-cell subpopulation expressing beta 7 integrin. REFERENCES 1. WHO RI. Vaccines and Biologicals 2016 available at https://www.who.int/immunization/ monitoring_surveillance/burden/estimates/rota virus/en/ 2. Johns Hopkins Bloomberg School of Public Health International Vaccine Access Center (IVAC): https://view-hub.org/map/? set=current-vaccine-intro status&category= rv&group=vaccine-introduction 3. Madhi SA, Cunliffe NA, Steele D, Witte D, Kirsten M, Louw C, Ngwira B, Victor JC, Gillard PH, Cheuvart BB, Han HH, Neuzil KM Malawi. Effect of human rotavirus vaccine on severe diarrhea in African infants. Med J 2016 Sep; 28(3):108-114. 4. Zaman K, Sack DA, Neuzil KM, Yunus M, Moulton LH, Sugimoto JD, Fleming JA, Hossain I, Arifeen SE, Azim T, Rahman M, Lewis KDC, Feller AJ, Qadri F, Halloran ME, Cravioto A, Victor JC PLoS. Effectiveness of a Journal OF MILITARY PHARMACO - MEDICINE N04 - 2021 161 live oral human rotavirus vaccine after programmatic introduction in Bangladesh: A cluster-randomized trial. Med. 2017 Apr. 5. Bhandari N, Rongsen-Chandola T, Bavdekar A, John J, Antony K, Taneja S, Goyal N, Kawade A, Kang G, Rathore SS, Juvekar S, Muliyil J, Arya A, Shaikh H, Abraham V, Vrati S, Proschan M, Kohberger R, Thiry G, Glass R, Greenberg HB, Curlin G, Mohan K, Harshavardhan GV, Prasad S, Rao TS, Boslego J, Bhan MK, India Rotavirus Vaccine Group. Efficacy of a monovalent human-bovine (116E) Rotavirus vaccine in Indian infants: A randomised, double-blind, placebo-controlled trial. Lancet 2014 Jun 21; 383(9935):2136-2143. 6. Dotan I, Allez M, Danese S, Keir M, Tole S, McBride, J. (2020). The role of integrins in the pathogenesis of inflammatory bowel disease: Approved and investigational anti-integrin therapies. Medicinal Research Reviews, 40(1), 245–262. https://doi.org/10.1002/med.21601. 7. Results of report on the pre-acceptance test of subjects of the Ministry of Health: Immunogenicity, safety and reactogenicity of ROTAVAC® in healthy infants aged 6 - 8 weeks in Viet Nam. 8. Mora JR, Von Andrian UH. Retinoic acid: An educational "vitamin elixir" for gut-seeking T cells. Immunity 2004 Oct; 21(4):458-60. doi: 10.1016/j.immuni.2004.10.002. PMID: 15485623. 9. Duester G. Families of retinoid dehydrogenases regulating vitamin A function: Production of visual pigment and retinoic acid. Eur J Biochem 2000 Jul; 267(14):4315-24. doi: 10.1046/j.1432-1327.2000.01497.x. PMID: 10880953.
File đính kèm:
 a_survey_on_some_immune_indicators_in_the_group_of_vietnames.pdf
a_survey_on_some_immune_indicators_in_the_group_of_vietnames.pdf

