Chrysin flavonoid adsorbed on B12N12 nanocage - A novel antioxidant nanomaterial
Antioxidative activity of chrysin (CYS) on the B12N12 nanocage has been evaluated by density functional theory
with B3PW91-D3 and M06-2X-D3 methods. Adsorption behavior and study of topologies demonstrated that the CYS
has chemisorbed to the nanocage and shows notable changes in the electronic properties of B12N12. The antioxidant
properties of the CYS and CYS/B12N12 systems have been studied in the different environments by the M06-2X-D3
method. The findings demonstrated that in the vacuum phase and water, benzene, and ethanol solvents, the BDE (5OH), PDE, PA values of CYS/B12N12 are smaller than those of CYS system. The current study implied that B12N12
nanocage can increase the antioxidative properties of the CYS

Trang 1

Trang 2

Trang 3

Trang 4

Trang 5
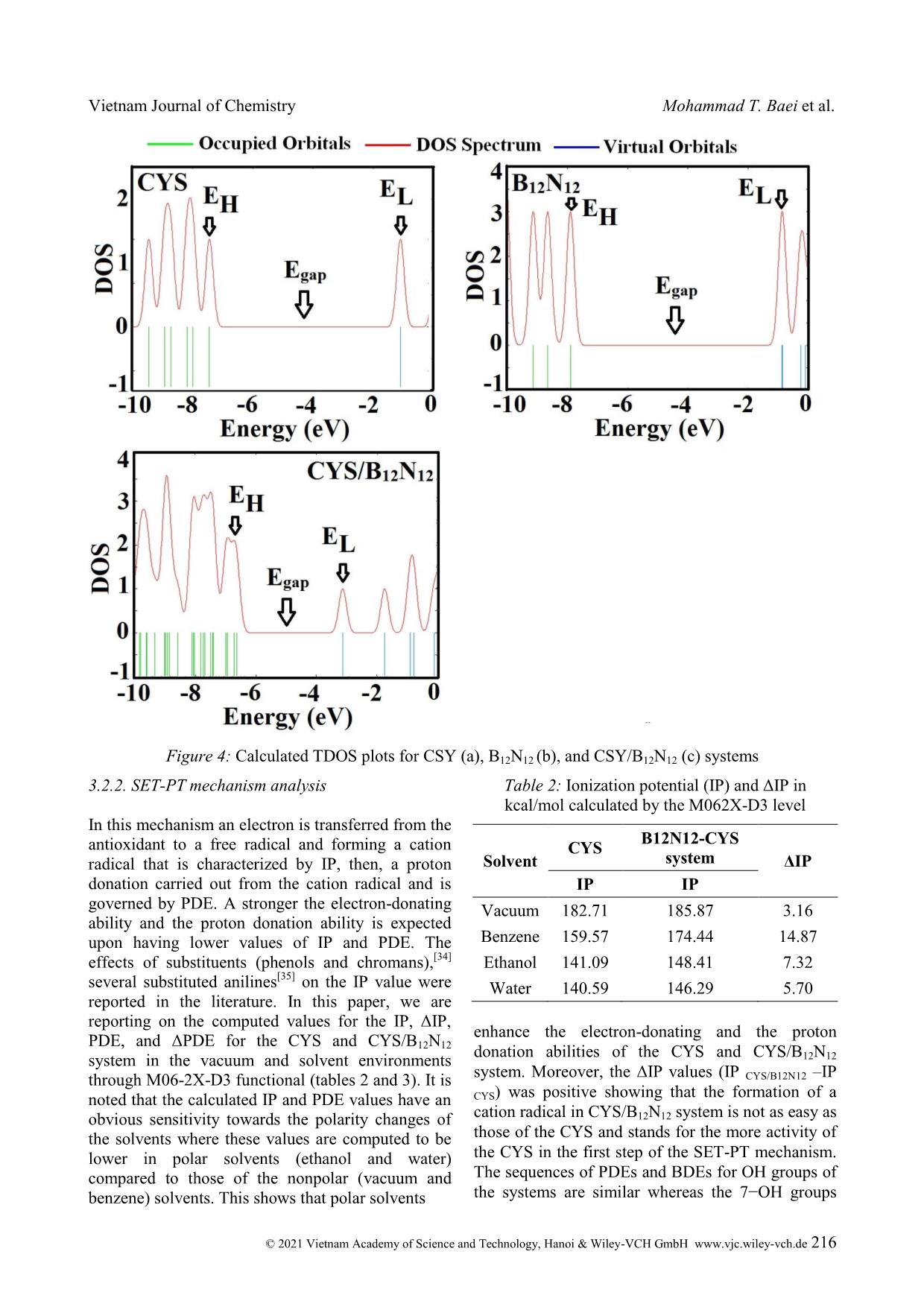
Trang 6

Trang 7

Trang 8

Trang 9

Trang 10
Tóm tắt nội dung tài liệu: Chrysin flavonoid adsorbed on B12N12 nanocage - A novel antioxidant nanomaterial

Cite this paper: Vietnam J. Chem., 2021, 59(2), 211-220 Article DOI: 10.1002/vjch.202000168 211 Wiley Online Library © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH Chrysin flavonoid adsorbed on B12N12 nanocage - A novel antioxidant nanomaterial Atefeh Khalili 1 , Mohammad T. Baei 2* , Seyed Hossein Hosseini Ghaboos 1 1 Department of Food Science and Technology, Azadshahr Branch, Islamic Azad University, Azadshahr, Golestan, Iran, postal code: 49617-89985 2 Department of Chemistry, Azadshahr Branch, Islamic Azad University, Azadshahr, Golestan, Iran Submitted September 29, 2020; Accepted January 8, 2021 Abstract Antioxidative activity of chrysin (CYS) on the B12N12 nanocage has been evaluated by density functional theory with B3PW91-D3 and M06-2X-D3 methods. Adsorption behavior and study of topologies demonstrated that the CYS has chemisorbed to the nanocage and shows notable changes in the electronic properties of B12N12. The antioxidant properties of the CYS and CYS/B12N12 systems have been studied in the different environments by the M06-2X-D3 method. The findings demonstrated that in the vacuum phase and water, benzene, and ethanol solvents, the BDE (5O- H), PDE, PA values of CYS/B12N12 are smaller than those of CYS system. The current study implied that B12N12 nanocage can increase the antioxidative properties of the CYS. Keywords. Chrysin, antioxidative activity, antiradical mechanisms, B12N12, DFT. 1. INTRODUCTION Flavonoids have been accepted as one of the largest and most widespread bioactive materials, and subset of phenolic compounds that can be found in vegetables, plants, and fruits. [1] Flavonoids showed potent scavenger activity against reactive nitrogen and oxygen species. They can transfer hydrogen’s and electrons to RONS which stabilizes them providing relatively permanent flavonoid radicals. Furthermore, flavonoids can chelate to metals for the prevention of radicals generation as well as activating antioxidant enzymes in deactivating free radicals. They are used in food products of the packaging in order to enhance the products' shelf-life and bioactive compound content due to their oxygen-sensitivity as an active antioxidant material. [2] Chrysin is a flavonoid and an analog of apigenin included in natural products (Pleurotus ostreatus, [3] propolis, [4] honey, [5] etc.) and many plants (Passiflora caerulea, [6] Passiflora incarnate, [7] Oroxylum indicum, [8] etc.). It has the high remedial power of transferring the intestinal membrane and also can be used to afford a wide variety of pharmacological activities particularly anti-inflammatory and antioxidant [8] properties. In late years, there has been an increasing attachment in using boron nitride nanotubes and other boron nitride nanostructures as promising materials for therapeutic agents [9-10] with significant prominence in cancer therapy. Boron nitride (BN) has distinguishing features containing substantial electrical-insulating performance, high resistance to oxidation, high Young’s modulus high thermal conductivity and stability, and high chemical inertness. [11] BN fullerenes were characterized by electron irradiation or arc-melting methods, with their chemical compositions and cage-like structures were examined by transmission electron microscopy (TEM) and time-of-flight mass spectrometry (TOFMS). [12] BNNPs have become an important topic in this field because of the wide availability of boron nitride and its inherent features of low toxicity, biodegradability and biocompatibility. [13] Also, in the last few decades, the potential of boron nitride for biomedical uses in the medic field, such as drug delivery, imaging and cellule stimulation was increased. [14] Hence, adsorption of chrysin on appropriate surfaces can be used as an election to increase its lifetime. There are several kinds of research focused on the chrysin adsorption on different substrates. [15] Moreover, boron nitride nanostructures have been widely used for the detection and sorption of drugs. [16,17] On the other hand, in late years, research into boron-including compounds has notably increased in pharmaceutical chemistry. [18] Also, it has been widely used for the detection and absorption of noble gases. [19] It Vietnam Journal of Chemistry Mohammad T. Baei et al. © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 212 is, therefore, significant to comprehend the influence of B12N12 nanocage on the antiradical activity of chrysin in order to deliver new clues to the development of antioxidants. Moreover, we evaluated the efficacy of the polar and non-polar solvents on the antioxidative activity of the systems. 2. COMPUTATIONAL METHODS We evaluated the improvement of antiradical activity of the CYS through the interaction with B12N12 nanocage by DFT calculations. Geometries, charge transfer characteristics (QT) between CYS and the nanocage, density of states (DOS), molecular electrostatic potential (MEP), and frontier molecular orbital (FMO) of the considered systems are computed with B3PW91- GD3BJ and M06-2X- D3 methods. The M06-2X method [20] is usually used to be the most appropriate one for main groups in chemistry and noncovalent interactions. [21] Therefore, geometry calculations and vibration frequencies were carried out on the nanocage, all molecules, ions, and various chrysin/B12N12 systems by the M06-2X/6-31G* [22,23] method with an empirical dispersion term (M06-2X-D3) in the Gaussian 09 program. [24] Then, the vibrational frequencies for the optimized geometries were computed at the M06-2X-D3 level combined with the 6-311+G* basis set for thermodynamic parameters. For the systems, the basis ... ex. These findings suggest that the interaction of CYS on the B12N12 nanocage enhances the antiradical activity of the CYS. The ETE values of the CYS were obtained to be smaller than that the CYS/B12N12 complex (ΔETE is positive) meaning that the CYS is more active than the CYS/B12N12 system in the second step of the SPLET method. Table 4: Proton affinity (PA) and ΔPA in kcal/mol calculated by the M062X/6-311+G* method Solvent PA ΔPA CYS B12N12-CYS system 5O-H 7O-H 5O-H 7O-H 5O-H 7O-H Vacuum 351.84 334.40 321.19 309.41 -30.65 -24.99 Benzene 113.89 98.62 91.09 82.93 -22.80 -15.69 Ethanol 49.29 41.75 38.86 35.68 -10.43 -6.07 Water 50.24 43.96 41.35 38.94 -8.89 -5.02 3.2.4. Thermodynamically preferred mechanism of the investigated structures The mechanism of this process is separated into two steps which the first step is substantially based on the thermodynamic aspect. Normally, the mechanisms of HAT, SET-PT and SPLET are dependent on the BDE, IP, and PA parameters whereas these parameters are generally implemented in defining the thermodynamically preferred reaction pathway in the free radical scavenging reactions. Tables 1, 2, and 4 of the vacuum and benzene environments represent that the lowermost BDEs are smaller than PA and the lowest IP. Herein, the free radical scavenging progress of the CYS and CYS on the B12N12 nanocage preferably and most possibly Vietnam Journal of Chemistry Mohammad T. Baei et al. © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 218 proceed through the HAT mechanism in these mentioned environments. Moreover, the order of BDE, IP, and PA are as follows: PA < BDE < IP in ethanol and water environments. Consequently, SPLET mechanism is the most desirable route for the free radical scavenging progress of the CYS and, CYS by B12N12 nanocage in these studied systems. 3.2.5. The antiradical activity influenced by the B12N12 nanocage on the CYS The result of our calculations demonstrates that in the vacuum and the solvent environments, the values of IP and ETE for the CYS are dropped slightly than those of CYS/B12N12 system (tables 2 and 5). In other words, the obtained results represents that in the different environments, the BDE (5O-H), PDE, PA values of CYS/B12N12 are smaller than that of CYS system. Therefore, the CYS adsorption on B12N12 surface can improve the antiradical activity of the CYS. Table 5: Electron transfer enthalpy (ETE) and ΔETE in kcal/mol calculated by the M062X/6-311+G* method Solvent ETE Δ ETE CYS B12N12-CYS system 5O-H 7O-H 5O-H 7O-H 5O-H 7O-H Vacuum 61.41 67.82 90.04 84.51 28.63 16.69 Benzene 83.45 89.65 103.18 108.81 19.73 19.16 Ethanol 107.90 112.75 116.17 120.70 8.27 7.95 Water 111.46 115.83 118.33 121.08 6.87 5.25 4. CONCLUSIONS In summary, the adsorption behavior and antioxidative activities of CYS and CYS/B12N12 systems have been successfully evaluated in vacuum, water, ethanol, and benzene environments. The result showed that CYS can chemisorbed via the C=O bond to a boron atom of the nanocage with Eads of -32.16 and -31.88 kcal/mol at the B3PW91-D/6- 31G* and M06-2X-D/6-31G* methods in vacuum environment, respectively. The results presented that adsorption of the CYS on the B12N12 surface induces remarkable changes in electronic properties of the nanocage and its Egap is diminished after adsorption process. In fact, the CYS can improve the electronic properties of the B12N12 surface by CYS adsorption and can thus create this nanocage more reactive. Besides, the adsorption of CYS on the B12N12 surface plays a significant role in the antioxidative activity of CYS. Therefore, in this research, M062X method was used to study the influence of the CYS adsorption on the B12N12 nanocage on the antioxidative activity of CYS based on HAT, SET- PT and SPLET methods. For this objective, values of the BDE, IP, PDE, PA, and ETE were evaluated in vacuum, ethanol, benzene, and water environments to better comprehend the antiradical progress of the studied systems. In the vacuum and benzene environments, except for compound CYS/B12N12 in benzene phase, BDEs values are smaller than the IP and PA. Therefore, in the phases, the antioxidative progress of the CYS and CYS/B12N12 complex undergoes the HAT mechanism with most possibility. In ethanol and water environments, sequences for BDE, IP and PA can be arranged in the following order: PA < BDE < IP. Therefore, in the environments, SPLET is the most remarkable method in the antioxidative progress of the CYS and CYS/B12N12 systems. In the vacuum and the solvent environments, the values of ETE and IP for the CYS are smaller than those of CYS/B12N12 system. In vacuum, benzene, ethanol, and water environments, the BDE (5O-H), PDE, PA values of CYS/B12N12 are smaller than that of CYS system. As a result, the adsorption of CYS on B12N12 nanocage would improve the antioxidative activity of the CYS. The outcome of this work indicates that the CYS adsorption on the B12N12 surface improves the antiradical activity of CYS and this fact may be effective in the progress of new antiradicals and also on the studies of the other structural characters on the antioxidative activity of flavonoids in future. Acknowledgments. This work was financially supported by Islamic Azad University, Azadshahr Branch. Conflict of interest. The authors declare that they have no conflict of interest. REFERENCES 1. J. B. Harborne. The Flavonoids: Advances in Research since 1986, Chapman and Hall, London, 1994. 2. Y. Fu, J. Chen, Y-J. Li, Y-F. Zheng, P. Li. Vietnam Journal of Chemistry Chrysin flavonoid adsorbed on B12N12 nanocage © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 219 Antioxidant and anti-inflammatory activities of six flavonoids separated from licorice, Food Chem., 2013, 141, 1063-1071. 3. R. Anandhi, T. Annadurai, T. S. Anitha, A. R. Muralidharan, K. Najmunnisha, V. Nachiappan, P. A. Thomas, P. Geraldine. Antihypercholesterolemic and antioxidative effects of an extract of the oyster mushroom, Pleurotus ostreatus, and its major constituent, chrysin, in Triton WR-1339-induced hypercholesterolemic rats, J. Physiol. Biochem., 2013, 69(2), 313-323. 4. M. Debiaggi, F. Tateo, L. Pagani, M. Luini, E. Romero. Effects of propolis flavonoids on virus infectivity and replication, Microbiologica, 1990, 13(3), 207-213. 5. I. Martos, F. Ferreres, F. A. Tomas-Barberan. Identification of flavonoid markers for the botanical origin of Eucalyptus honey, Journal of Agricultural and Food Chemistry, 2000, 48(5), 1498-1502. 6. C. Wolfman, H. Viola, A. Paladini, F. Dajas, J. H. Medina. Possible anxiolytic effects of chrysin, a central benzodiazepine receptor ligand isolated from Passiflora coerulea, Pharmacology, Biochemistry and Behavior, 1994, 47(1), 1-4. 7. E. Brown, N. S. Hurd, S. McCall, T. E. Ceremuga. Evaluation of the anxiolytic effects of chrysin, a Passiflora incarnate extract, in the laboratory rat, AANA Journal, 2007, 75(5), 333-337. 8. Harminder, V. Singh, A. K. Chaudhary. A review on the taxonomy, ethnobotany, chemistry and pharmacology of Oroxylum indicum vent, Indian Journal of Pharmaceutical Sciences, 2011, 73(5) 483-490. 9. S. Feng, H. Zhang, C. Zhi, X.-D. Gao, H. Nakanishi. pH-responsive charge-reversal polymer- functionalized boron nitride nanospheres for intracellular doxorubicin delivery, Int. J. Nanomed., 2018, 13, 641-652. 10. X. Li, C. Zhi, N. Hanagata, M. Yamaguchi, Y. Bando, D. Golberg. Boron nitride nanotubes functionalized with mesoporous silica for intracellular delivery of chemotherapy drugs, Chem. Commun., 2013, 49, 7337-7339. 11. D. Golberg, Y. Bando, Y. Huang, T. Terao, M. Mitome, C. Tang, C. Zhi. Boron nitride nanotubes and nanosheets, ACS Nano, 2010, 4, 2979-2993. 12. O. Ste´phan, Y. Bando, A. Loiseau, F. Willaime, N. Shramchenko, T. Tamiya, T. Sato. Formation of small single-layer and nested BN cages under electron irradiation of nanotubes and bulk material, Appl. Phys. A, 1998, 67, 107-111. 13. G. Ciofani, S. Danti, D. D’Alessandro, S. Moscato, A. Menciassi. Assessing cytotoxicity of boron nitride nanotubes: Interference with the MTT assay, Biochem. Biophys. Res. Commun., 2010, 394, 405- 411. 14. A. Merlo, V. R. S. S. Mokkapati, S. Pandit, I. Mijakovic. Boron nitride nanomaterials: biocompatibility and bio-applications, Biomater. Sci., 2018, 6, 2298-2311. 15. G. Sathishkumar, R. Bharti, P. K. Jha, M. Selvakumar, G. Dey, R. Jha, M. Jeyaraj, M. Mandal, S. Sivaramakrishnan. Dietary flavone chrysin (5,7- dihydroxyflavone ChR) functionalized highly-stable metal nanoformulations for improved anticancer applications, RSC Adv., 2015, 5, 89869-89878. 16. M. Bezi Javan, A. Soltani, E. Tazikeh Lemeski, A. Ahmadi, S. Moazen Rad. Interaction of B12N12 nano- cage with cysteine through various functionalities: a DFT study, Superlattice Microstruct., 2016, 100, 24- 37. 17. A. Soltani, A. Sousaraei, M. Bezi Javan, M. Eskandari, H. Balakheyli. Electronic and optical properties of 5-AVA functionalized BN nanocages: a DFT study, New J. Chem., 2016, 40, 7018-7026. 18. E. Andrade-Jorge, A. K. Garcia-Avila, A. L. Ocampo-Nestor, J. G. Trujillo-Ferrara, M. A. Soriano-Ursua. Advances of bioinformatics applied to development and evaluation of boron-containing compounds, Curr. Org. Chem., 2018, 22, 298-306. 19. R. Pino-Rios, E. Chigo-Anota, E. Shakerzadeh, G. Cárdenas-Jirón. B12N12 cluster as a collector of noble gases: A quantum chemical study, Phys. E (Amsterdam, Neth.), 2020, 115, 113697. 20. Y. Zhao, D. G. Truhlar. Hybrid Meta Density Functional Theory Methods for Thermochemistry, Thermochemical Kinetics, and Noncovalent Interactions: The MPW1B95 and MPWB1K Models and Comparative Assessments for Hydrogen Bonding and van der Waals Interaction, J. Phys. Chem. A, 2004, 108, 6908. 21. Y. Zhao, D. G. Truhlar. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals, Theor. Chem. Acc., 2008, 120, 215. 22. R. C. Binning Jr, L. A. Curtiss. Compact contracted basis sets for third-row atoms: Ga-Kr, J. Comput. Chem., 1990, 11, 1206-1216. 23. J. P. Blaudeau, M. P. McGrath, L. A. Curtiss, L. Radom. Extension of Gaussian-2 (G2) theory to molecules containing third-row atoms K and Ca, J. Chem. Phys., 1997, 107, 5016-5021. 24. M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, et al., Gaussian, Inc., Wallingford CT, 2009. 25. A. V. Marenich, C. J. Cramer, D. G. Truhlar. Universal Solvation Model Based on Solute Electron Density and on a Continuum Model of the Solvent Defined by the Bulk Dielectric Constant and Atomic Surface Tensions, J. Phys. Chem. B, 2009, 113, 6378- 6396. Vietnam Journal of Chemistry Mohammad T. Baei et al. © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 220 26. Z. Marković, D. Milenković, J. Đorović, J. M. D. Marković, V. Stepanić, B. Lučić, D. Amić. PM6 and DFT study of free radical scavenging activity of morin, Food Chem., 2012, 134, 1754-1760. 27. K. Jomova, M. Lawson, L. Drostinova, P. Lauro, P. Poprac, V. Brezova et al. Protective role of quercetin against copper(II)-induced oxidative stress: a spectro- scopic, theoretical and DNA damage study. Food Chem. Toxicol., 2017, 110, 340-350. 28. Y. Zheng, G. Deng, D. -F. Chen, R. Guo, R. C. Lai. The influence of C2 double bond C3 double bond on the antiradical activity of flavonoid: Different mechanisms analysis, Phytochemistry, 2019, 157, 1- 7. 29. J. Rimarclk, V. Lukes, E. Klein, M. Ilcin. Study of the solvent effect on the enthalpies of homolytic and heterolytic N–H bond cleavage in p- phenylenediamine and tetracyano-p- phenylenediamine, J. Mol Struc, THEOCHEM, 2010, 952, 25-30. 30. V. D. Parker. Homolytic bond (H-A) dissociation free energies in solution. Applications of the standard potential of the (H+/H.bul.) couple, J. Am. Chem. Soc., 1992, 114, 7458-7462. 31. G. Sathishkumar, R. Bharti, P. K. Jha, M. Selvakumar, G. Dey, R. Jha, M. Jeyaraj, M. Mandal, S. Sivaramakrishnan. Dietary flavone chrysin (5,7- dihydroxyflavone ChR) functionalized highly-stable metal nanoformulations for improved anticancer applications, RSC Adv., 2015, 5, 89869-89878. 32. J. Cano Ordaz, E. Chigo Anota, M. Salazar Villanueva, M. Castro. Possibility of a magnetic [BN fullerene: B6 cluster]-nanocomposite as a vehicle for the delivery of dapsone, New J. Chem., 2017, 41, 8045-8052. 33. N. Gerayeli, M. Tafazzoli, M. Ghiasi. Theoretical Study on Glycosyl Group Effect on Antioxidant Ability of Chrysin Bioflavonoid, Phys. Chem. Res., 2016, 4, 643-654. 34. E. Klein, V. Lukes. DFT/B3LYP Study of the Substituent Effect on the Reaction Enthalpies of the Individual Steps of Single Electron Transfer-Proton Transfer and Sequential Proton Loss Electron Transfer Mechanisms of Phenols Antioxidant Action, J. Phys. Chem. A, 2006, 110, 12312-12320. 35. E. Klein, J. Rimarcik, V. Lukes. DFT/B3LYP Study of the O–H Bond Dissociation Enthalpies and Proton Affinities of para- and meta-Substituted Phenols in Water and Benzene, Acta Chim. Slov., 2009, 2, 37- 51. Corresponding author: Mohammad T. Baei Department of Chemistry Azadshahr Branch, Islamic Azad University, Azadshahr, Golestan, Iran E-mail: Baei52@yahoo.com.
File đính kèm:
 chrysin_flavonoid_adsorbed_on_b12n12_nanocage_a_novel_antiox.pdf
chrysin_flavonoid_adsorbed_on_b12n12_nanocage_a_novel_antiox.pdf

