Tổng quan về lignin: Cấu trúc, phương pháp tổng hợp và ứng dụng
Lignin là polymer tự nhiên phong phú thứ hai trên thế giới, sau cellulose. Nó thường được tạo ra từ sản phẩm phụ của
các ngành công nghiệp sản xuất giấy và ethanol. Với sự phức tạp và đa dạng của các nhóm chức trong cấu trúc, lignin
trở nên hấp dẫn để được chuyển đổi thành nhiều loại sản phẩm có giá trị cao như khí tổng hợp, sợi carbon, nhựa
phenolic, các sản phẩm oxy hóa và hydro-cacbon đa chức năng. Mục đích của nghiên cứu này là cung cấp một cái nhìn
tổng quan toàn diện về cấu trúc và các loại lignin khác nhau, cũng như những tiến bộ gần đây trong kỹ thuật thu nhận
lignin. Bên cạnh đó, những ứng dụng tiềm năng, định hướng phát triển trong tương lai của lignin cũng được thảo luận
chi tiết.

Trang 1

Trang 2
Trang 3

Trang 4

Trang 5

Trang 6
Tóm tắt nội dung tài liệu: Tổng quan về lignin: Cấu trúc, phương pháp tổng hợp và ứng dụng

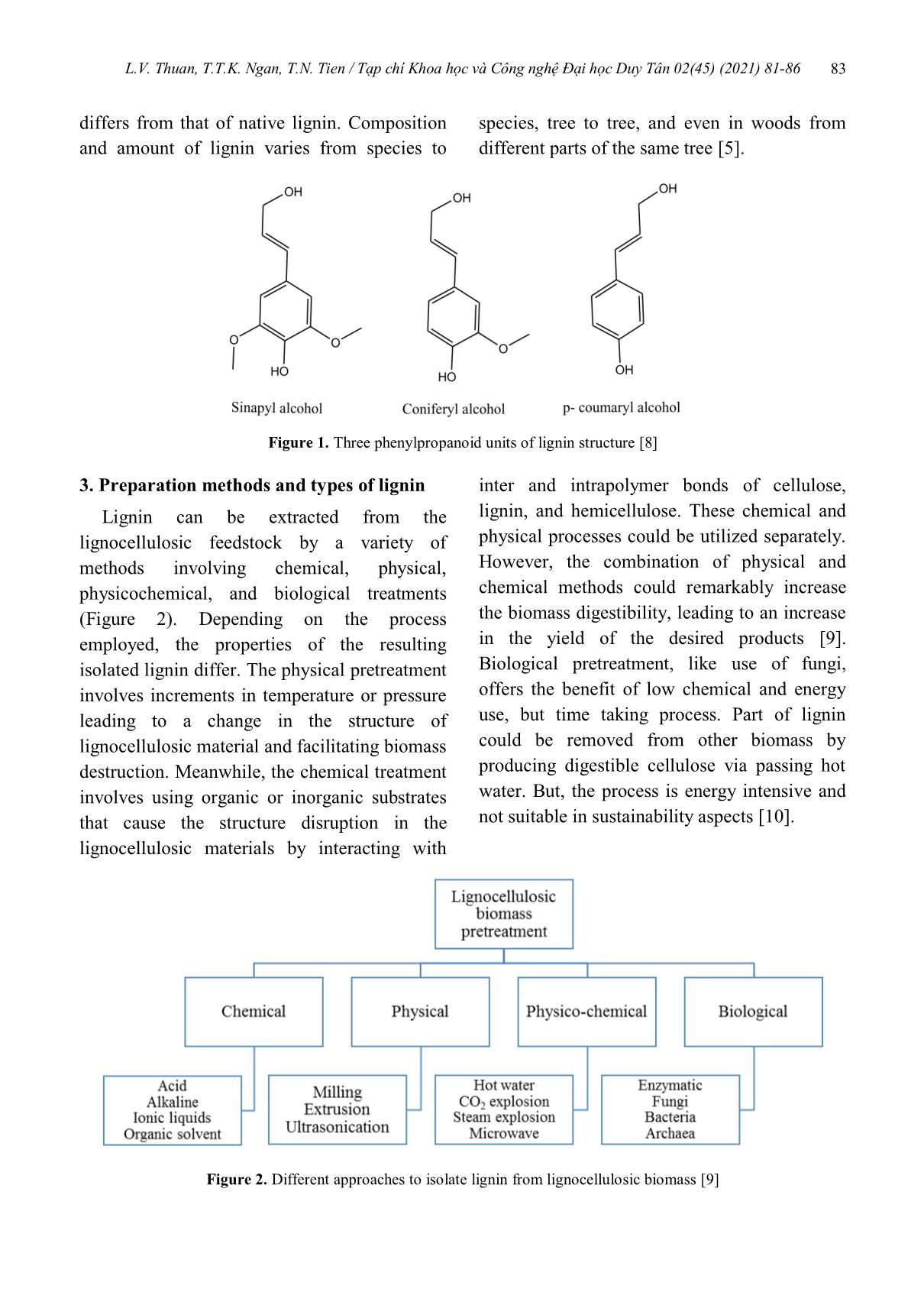
L.V. Thuan, T.T.K. Ngan, T.N. Tien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 81-86 81 A mini review on lignin: Structures, preparations, and applications Tổng quan về lignin: Cấu trúc, phương pháp tổng hợp và ứng dụng Le Van Thuana,b, Tran Thi Kieu Nganb, Tran Nguyen Tiena,b* Lê Văn Thuậna,b, Trần Thị Kiều Ngânb, Trần Nguyên Tiếna,b* aCenter for Advanced Chemistry, Institute of Research and Development, Duy Tan University, Da Nang City, 550000, Vietnam aTrung tâm Hóa học Tiên tiến, Viện Nghiên cứu và Phát triển Công nghệ Cao, Đại học Duy Tân, Đà Nẵng, Việt Nam bThe Faculty of Environmental and Chemical Engineering, Duy Tan University, Da Nang City, 550000, Vietnam bKhoa Môi trường và Công nghệ Hóa, Đại học Duy Tân, Đà Nẵng, Việt Nam (Ngày nhận bài: 03/03/2021, ngày phản biện xong: 09/03/2021, ngày chấp nhận đăng: 05/03/2021) Abstract Lignin is the second most abundant natural renewable biopolymer after cellulose on the earth. It is commonly generated as a by-product from the paper and ethanol industry. The complexity and richness of its functional groups make lignin attractive for converting into a variety of high-value products such as syngas, carbon fiber, phenolic resin, various oxidized products, and multifunctional hydro-carbons. This work intends to provide a comprehensive overview of structures and different types of lignin, as well as the recent progress in its preparation techniques. Besides, the extensive range of applications and opportunities of lignin were also discussed in detail. Keywords: Lignin, lignocellulosic biomass, extraction methods; lignin applications. Tóm tắt Lignin là polymer tự nhiên phong phú thứ hai trên thế giới, sau cellulose. Nó thường được tạo ra từ sản phẩm phụ của các ngành công nghiệp sản xuất giấy và ethanol. Với sự phức tạp và đa dạng của các nhóm chức trong cấu trúc, lignin trở nên hấp dẫn để được chuyển đổi thành nhiều loại sản phẩm có giá trị cao như khí tổng hợp, sợi carbon, nhựa phenolic, các sản phẩm oxy hóa và hydro-cacbon đa chức năng. Mục đích của nghiên cứu này là cung cấp một cái nhìn tổng quan toàn diện về cấu trúc và các loại lignin khác nhau, cũng như những tiến bộ gần đây trong kỹ thuật thu nhận lignin. Bên cạnh đó, những ứng dụng tiềm năng, định hướng phát triển trong tương lai của lignin cũng được thảo luận chi tiết. Từ khóa: Lignin, sinh khối lignocellulose, phương pháp tách chiết, ứng dụng của lignin 1. Introduction The lignin (lignum) is a component of lignocellulose which consists also of cellulose and hemicellulose. This material is the second most abundant natural polymer on earth, which plays an important role in plants, including providing mechanical supports, transporting water and minerals, and protecting plants or 02(45) (2021) 81-86 * Corresponding Author: Tran Nguyen Tien; Center for Advanced Chemistry, Institute of Research and Development, Duy Tan University, Da Nang City, 550000, Vietnam; The Faculty of Environmental and Chemical Engineering, Duy Tan University, Da Nang City, 550000, Vietnam Email: [email protected] L.V. Thuan, T.T.K. Ngan, T.N. Tien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 81-86 82 wood from chemical or microbial attacks [1].. Traditionally, lignin is considered as low-value waste product. However, it has been studied that lignin can be used to make high-value products such as syngas, carbon fiber, phenolic compounds, and multifunctional hydro-carbons [2]. In addition, due to its diverse reaction sites, high carbon content and low content of oxygen, lignin can become a potential sustainable alternative source of energy and chemicals [3]. The molecular structure of lignin is highly dependent on the resources and extraction processes. Different types of lignin contain different functional groups and show different molecular weight and elemental composition. Therefore, the structure of lignin is extremely complicated and difficult to determine. However, it is generally accepted that lignin is three- dimensional macromolecule formed by the coupling of three phenylpropane units (empirical formula of C31H34O11): guaiacyl, syringyl, and p-hydroxyphenyl alcohol, which are formed through Shikimate and Cinnamate pathways [4]. Lignin can be obtained from a variety of natural sources, including woody biomass, agricultural residues, and energy crops. Lignocellulosic biomass is primarily comprised of cellulose (38–50%), hemicellulose (23–32%) and lignin (12–25%) components. The biomass reserves on the earth have been estimated to be approximately 1.85–2.4×1012 tons and about 20% of this amount of biomass is lignin. Besides the natural abundance, lignin is also present as a major byproduct of the pulp and paper industry. About 50–70 million tons of lignin is produced annually at pulp and paper facilities world-wide. It is estimated by 2030, this number will increase by 225 million tons per year as the annual production of lignin [2]. However, the majority is discarded as waste or burnt to recover heat and electricity, causing serious environmental pollution and resource waste. Only approximately 2% of the produced lignin is isolated and effectively used for various products such as dispersants, adhesives, surfactants, and fuel [1]. Finding new and high value-added applications of lignin recovered from pulping waste liquor is imperative, which has both economic and environmental benefit. Currently, increasing research focuses on developing different approaches to lignin extraction and converting it into value-added products. In this review, we provide a comprehensive overview of the structures, preparation methods of lignin, and their applications in different commercial fields. In addition, the challenges, and opportunities of lignin applications were discussed in this study. 2. Structures of lignin Lignin is a complex, amorphous, branched polyphenolic macromolecule with aromatic polymeric structure [5]. The structure of lignin varies based on the extraction process, and the presence of various functional groups. Lignin exhibits a high recalcitrance to chemical and biochemical depolymerization due to the existence of phenylpropanoid polymers, ether linkages (β–O–4) and a range of functional groups namely methoxy, aliphatic and aromatic hydroxy, benzyl alcohol, ether and non–cyclic benzyl ether and carbonyls [6]. Lignin has different functional groups such as: hydroxyl, methoxyl, carbonyl, and carboxyl, etc. Lignin has three basic types of monomers; coniferyl alcohol, sinapyl alcohol, and p-coumaryl alcohol, also known as monolignols (Figure 1). Peroxidase and laccase enzymes in the plant can cause the dehydrogenation of phenolic OH groups and generate intermediate free radicals from these lignin precursors [7]. The exact structure of lignin in its native form in plants is still unclear, as studies have concluded that the structure is modified during its isolation and L.V. Thuan, T.T.K. Ngan, T.N. Tien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 81-86 83 differs from that of native lignin. Composition and amount of lignin varies from species to species, tree to tree, and even in woods from different parts of the same tree [5]. Figure 1. Three phenylpropanoid units of lignin structure [8] 3. Preparation methods and types of lignin Lignin can be extracted from the lignocellulosic feedstock by a variety of methods involving chemical, physical, physicochemical, and biological treatments (Figure 2). Depending on the process employed, the properties of the resulting isolated lignin differ. The physical pretreatment involves increments in temperature or pressure leading to a change in the structure of lignocellulosic material and facilitating biomass destruction. Meanwhile, the chemical treatment involves using organic or inorganic substrates that cause the structure disruption in the lignocellulosic materials by interacting with inter and intrapolymer bonds of cellulose, lignin, and hemicellulose. These chemical and physical processes could be utilized separately. However, the combination of physical and chemical methods could remarkably increase the biomass digestibility, leading to an increase in the yield of the desired products [9]. Biological pretreatment, like use of fungi, offers the benefit of low chemical and energy use, but time taking process. Part of lignin could be removed from other biomass by producing digestible cellulose via passing hot water. But, the process is energy intensive and not suitable in sustainability aspects [10]. Figure 2. Different approaches to isolate lignin from lignocellulosic biomass [9] L.V. Thuan, T.T.K. Ngan, T.N. Tien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 81-86 84 Two major processes popular in the pulp and paper industries to separate cellulose from lignin, commonly known as kraft (generating kraft lignin, sometimes also called alkali lignin) and sulfite pulping (generating lignosulfonate). Kraft pulping is the major chemical pulping process, accounting for 85% of the total lignin production in the world [11]. The process is performed at a high pH, and about 90–95% of the lignin is dissolved into the black liquor. Kraft lignin is typically precipitated and recovered from black liquor by the addition of acidifying agents. Predominantly, the acidification is carried out by adding either mineral acid (e.g., sulfuric acid) or carbon dioxide, followed by filtering, washing, and drying for the recovery of Kraft lignin. The sulfite pulping process is conducted between a pH of 2–12, depending on the cationic composition of the pulping liquor. The process uses a heated aqueous solution of a sulphite or bisulfite salt with counter cations such as sodium, ammonium, magnesium, or calcium. Lignosulfonates, isolated lignins from the sulfite process, contain significant amounts of sulfur in the form of sulfonate groups. Since lignosulfonates are widely available, lignosulfonates were used in a wide range of applications, such as dispersants, flocculants, concrete additives, and composites [9]. Organosolv process is one of the most relevant methods, which is based on the solubilization of lignin using a mixture of different organic solvents and water as cooking liquor. It is also helpful in extracting highly homogeneous lignin, which enables its further valorization into value-added products [7]. Since the organosolv process is conducted in the absence of sulfur, it has recently been utilized more so than Kraft and sulfite pulping. Furthermore, the large-scale production of organosolv lignin is expected from the emerging cellulosic ethanol sectors, which offers significant opportunities for lignin valorization. 4. Applications The efforts in using lignin derivatives in more sophisticated applications are currently booming. Lack of toxicity and versatility of lignin creates several potential industrial application routes. Stringent regulations, bulk availability, cost efficiency and growing need for bio-based and renewable chemicals are high-value lignin properties [12]. Lignins are being used for the controlled release of fertilizers, modified for slow-release fertilizers and herbicides in agriculture, as a base for different materials application in the fields of bioplastics, (nano)composites and nanoparticles [13]. By considering unique attributes of lignin, such as its binding properties, products, such as adhesive for wood, pellets, foundry resins, and epoxy resins could be produced. Properties, such as hydrophobicity, antioxidant and thermal resistance, could also facilitate lignin use in thermoplastics, composites, and packaging. Furthermore, as adsorbents in solution, protective UV-absorbents, dispersants, to improve the saccharification of lignocelluloses in the production of biofuels, in electro-chemical applications and in environmentally friendly functionalization approach to extend the role of lignin for future biomass and biofuel applications [14]. By con- trolling the structure of lignin, other advanced applications could be developed, such as nano/microcapsules, nano/microporous materials, and lignin nanotubes, as a smart DNA delivery without possessing the cytotoxicity related to carbon nanotubes [15]. Significant scope for diverse applications (Figure 3) principally segmented as power/energy, macromolecules and aromatics. L.V. Thuan, T.T.K. Ngan, T.N. Tien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 81-86 85 Figure 3. Application fields of lignin [12] 5. Conclusions This review discussed the structures, extraction methods, and applications of lignin. Lignin is available in huge amounts as a by- product of the pulping process, and this will further increase as the lignocellulosic ethanol process gets commercialized. Grand amount of lignin is produced worldwide. However, only a small amount is applicable for further applications such as additives, dispersants, binders, or surfactants. The remaining part is burn and cause environmental problem. Many research efforts have been done in developing processes that could produce valuable lignin- derived compound. Recently, there have been more investigations conducted on using lignin to construct medical materials, electrochemical energy materials, and 3D printing composites. In spite of the increasing utilization of lignin- based biomaterials, the high value-added applications of lignin still face some challenges mainly due to its complicated and changeable macromolecular structure. We hope that in the future, the advancement of science and technology will overcome these obstacles, thereby further expanding the valuable applications of lignin. References [1] Q. Tang, Y. Qian, D. Yang, X. Qiu, Y. Qin, and M. Zhou, “Lignin-Based Nanoparticles: A Review on Their Preparations and Applications,” Polymers (Basel)., vol. 12, no. 11, p. 2471, Oct. 2020, doi: 10.3390/polym12112471. [2] D. S. Bajwa, G. Pourhashem, A. H. Ullah, and S. G. Bajwa, “A concise review of current lignin production, applications, products and their environment impact,” Ind. Crops Prod., vol. 139, no. February, p. 111526, 2019, doi: 10.1016/j.indcrop.2019.111526. L.V. Thuan, T.T.K. Ngan, T.N. Tien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 81-86 86 [3] I. Spiridon, “Extraction of lignin and therapeutic applications of lignin-derived compounds. A review,” Environ. Chem. Lett., vol. 18, no. 3, pp. 771–785, 2020, doi: 10.1007/s10311-020-00981-3. [4] A. Eraghi Kazzaz, Z. Hosseinpour Feizi, and P. Fatehi, “Grafting strategies for hydroxy groups of lignin for producing materials,” Green Chem., vol. 21, no. 21, pp. 5714–5752, 2019, doi: 10.1039/C9GC02598G. [5] L. D. Tolesa and M.-J. Lee, “Strategies for Lignin Pretreatment, Decomposition and Modification: A Review,” J. Bahan Alam Terbarukan, vol. 9, no. 1, pp. 01–20, 2020, doi: 10.15294/jbat.v9i1.23392. [6] V. K. Ponnusamy et al., “A review on lignin structure, pretreatments, fermentation reactions and biorefinery potential,” Bioresour. Technol., vol. 271, pp. 462–472, 2019, doi: 10.1016/j.biortech.2018.09.070. [7] M. P. Pandey and C. S. Kim, “Lignin Depolymerization and Conversion: A Review of Thermochemical Methods,” Chem. Eng. Technol., vol. 34, no. 1, pp. 29–41, 2011, doi: 10.1002/ceat.201000270. [8] E. P. Feofilova and I. S. Mysyakina, “Lignin: Chemical structure, biodegradation, and practical application (a review),” Appl. Biochem. Microbiol., vol. 52, no. 6, pp. 573–581, 2016, doi: 10.1134/S0003683816060053. [9] A. Eraghi Kazzaz and P. Fatehi, “Technical lignin and its potential modification routes: A mini- review,” Ind. Crops Prod., vol. 154, no. May, 2020, doi: 10.1016/j.indcrop.2020.112732. [10] P. Dey, P. Pal, J. D. Kevin, and D. B. Das, “Lignocellulosic bioethanol production: prospects of emerging membrane technologies to improve the process – a critical review,” Rev. Chem. Eng., vol. 36, no. 3, pp. 333–367, Apr. 2020, doi: 10.1515/revce-2018-0014. [11] H. Chen, Lignocellulose Biorefinery Engineering: Principles and Applications. 2015. [12] N. Mandlekar et al., “An Overview on the Use of Lignin and Its Derivatives in Fire Retardant Polymer Systems,” in Lignin - Trends and Applications, 2018. [13] M. Norgren and H. Edlund, “Lignin: Recent advances and emerging applications,” Curr. Opin. Colloid Interface Sci., vol. 19, no. 5, pp. 409–416, 2014, doi: 10.1016/j.cocis.2014.08.004. [14] O. Yu and K. H. Kim, “Lignin to materials: A focused review on recent novel lignin applications,” Appl. Sci., vol. 10, no. 13, 2020, doi: 10.3390/app10134626. [15] S. M. R. Wahba, A. S. Darwish, I. H. Shehata, and S. S. Abd Elhalem, “Sugarcane bagasse lignin, and silica gel and magneto-silica as drug vehicles for development of innocuous methotrexate drug against rheumatoid arthritis disease in albino rats,” Mater. Sci. Eng. C, 2015, doi: 10.1016/j.msec.2014.12.054.
File đính kèm:
 tong_quan_ve_lignin_cau_truc_phuong_phap_tong_hop_va_ung_dun.pdf
tong_quan_ve_lignin_cau_truc_phuong_phap_tong_hop_va_ung_dun.pdf

