Sulfanilic acid catalysed one-Pot three-component Mannich reaction for synthesis of β-amino ketones
We have reported sulfanilic acid as an exceedingly competent catalyst for one-pot Mannich reaction to give β-
amino carbonyl compounds in good to excellent yield within a short reaction time. The various organic acids have
screened; like gallic acid, 4-hydroxy benzoic acid, 4-amino benzoic acid, phenylacetic acid, chloroacetic acid,
sulfosalicylic acid, sulfanilic acid, chloro benzoic acid, phthalic acid, salicylic acid, cinnamic acid, hippuric acid, 1-
naphthyl acetic acid, o-amino benzoic acid, p-TSA, succinic acid, malic acid, and among them sulfanilic acid is a
suitable catalyst. The reaction condition was optimized with respect to the solvent, the amount of catalyst as well as the
variation of the ketone, aldehyde, and amine substrates. The procedure is mild, effective, ecofriendly, and the use of the
minimum amount of catalyst.

Trang 1

Trang 2

Trang 3

Trang 4

Trang 5

Trang 6
Trang 7
Trang 8
Trang 9
Trang 10
Tải về để xem bản đầy đủ
Tóm tắt nội dung tài liệu: Sulfanilic acid catalysed one-Pot three-component Mannich reaction for synthesis of β-amino ketones

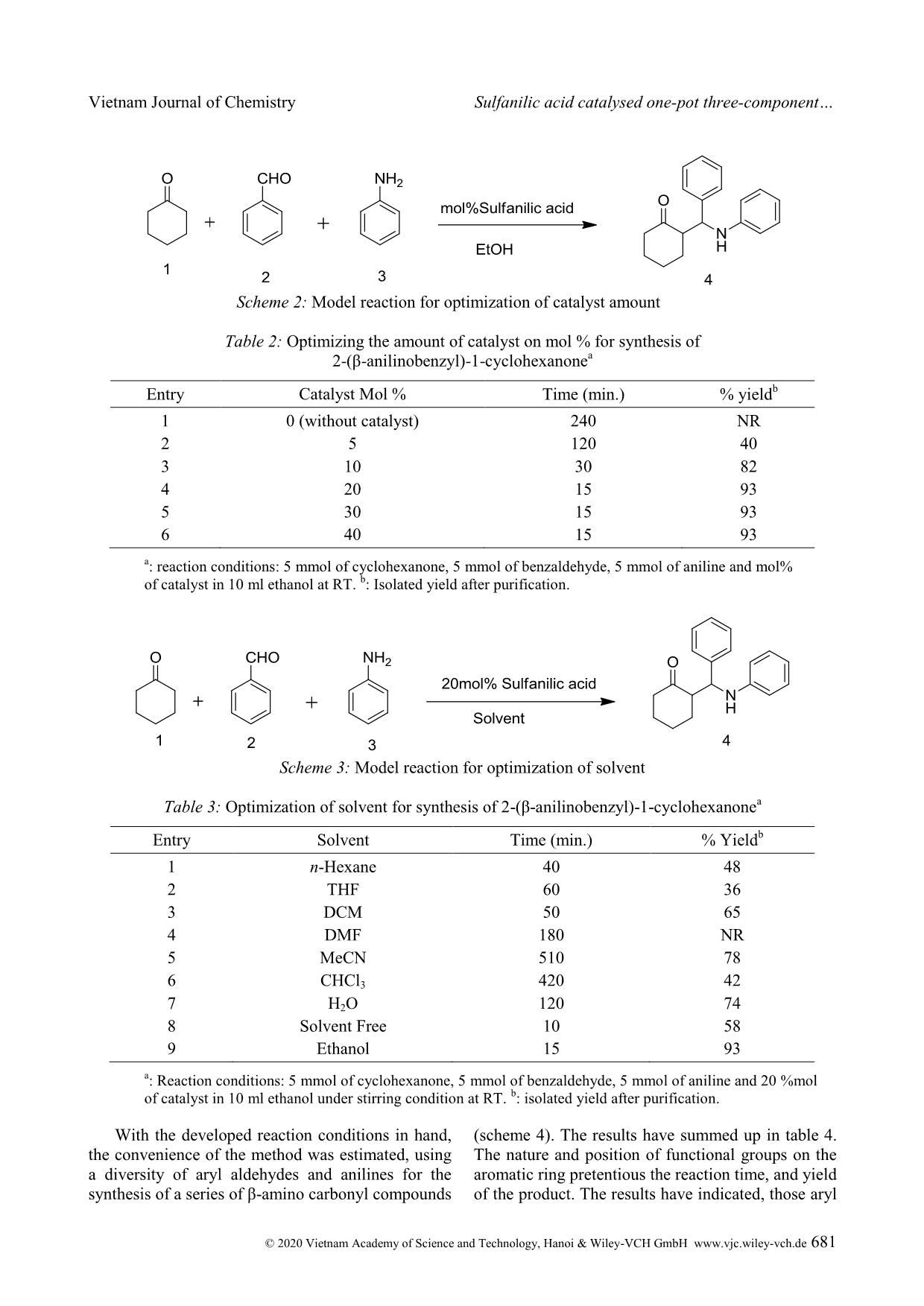
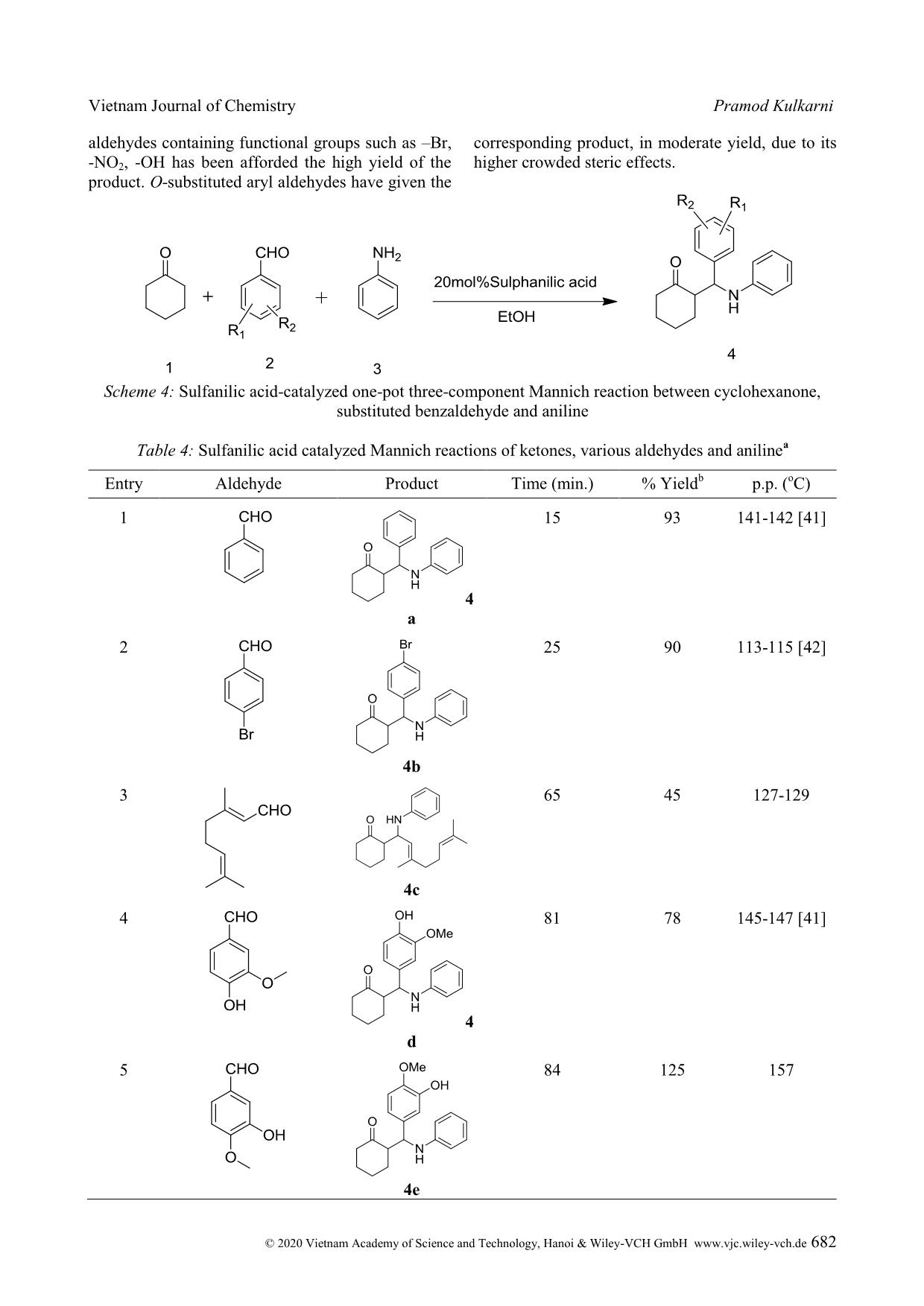
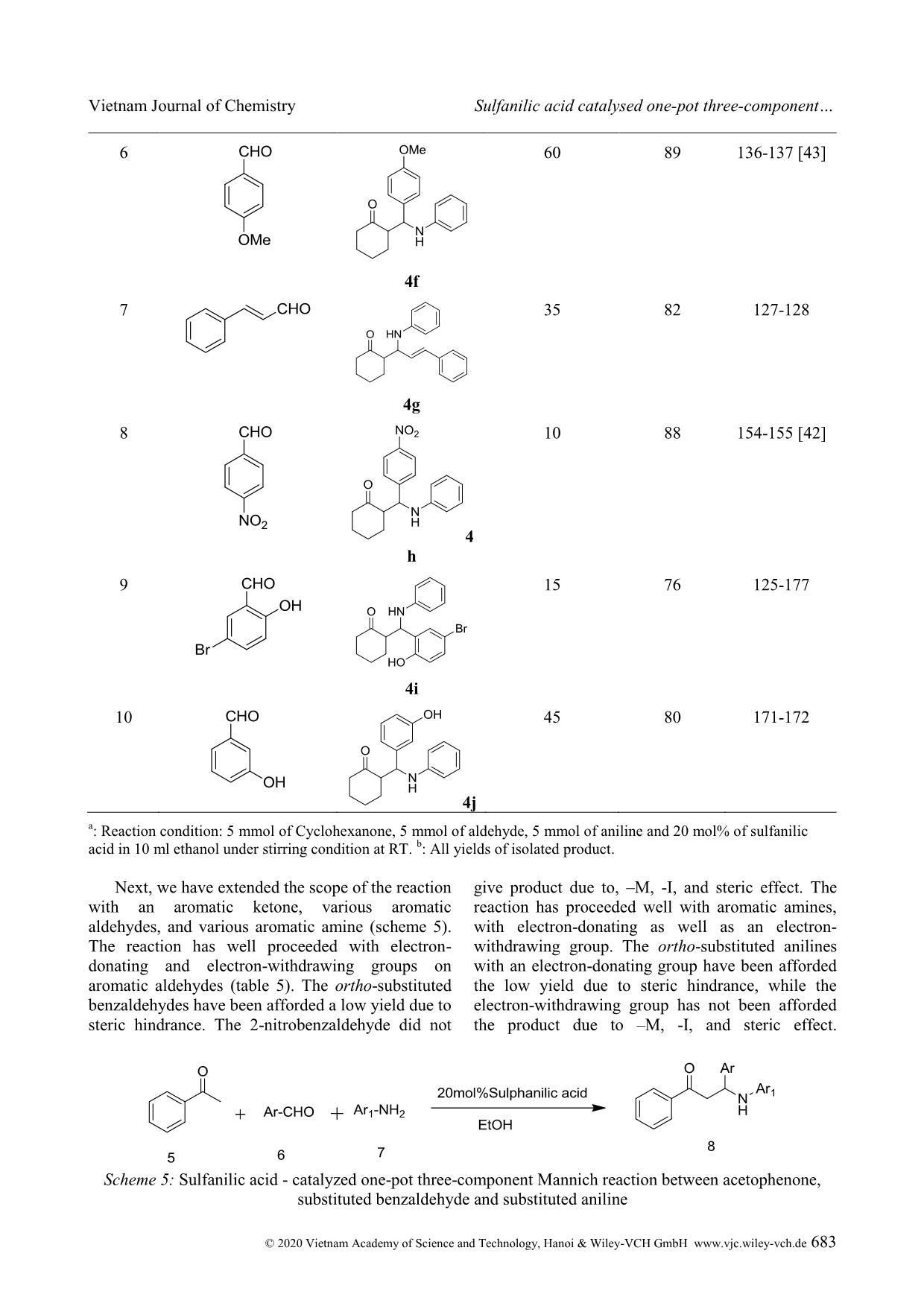
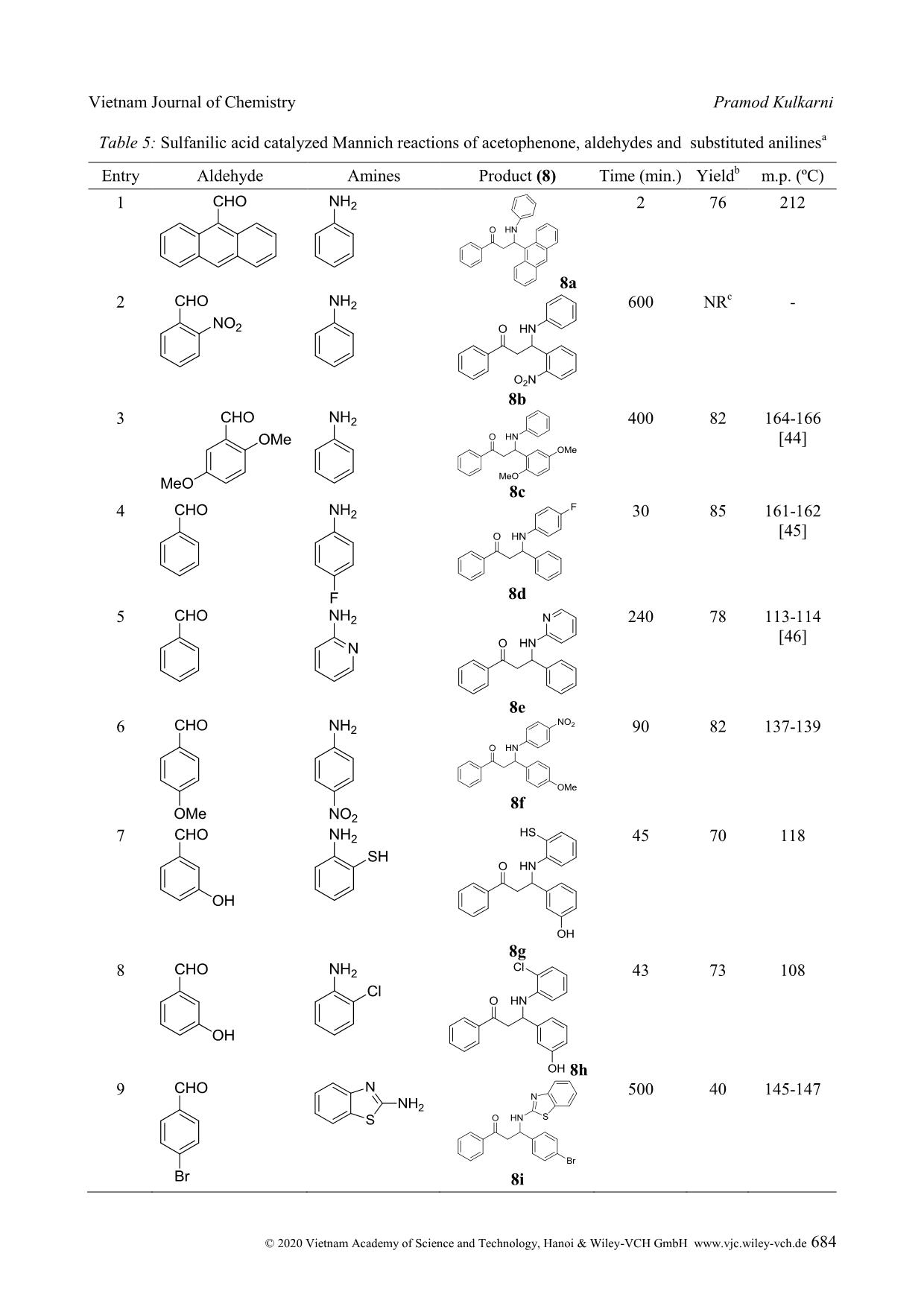
Cite this paper: Vietnam J. Chem., 2020, 58(5), 675-687 Article DOI: 10.1002/vjch.202000090 675 Wiley Online Library © 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH Sulfanilic acid catalysed one-pot three-component Mannich reaction for synthesis of β-amino ketones Pramod Kulkarni Department of Chemistry, Hutatma Rajguru Mahavidyalaya, Rajgurunagar, Pune-410505, MS India Submitted June 1, 2020; Accepted August 18, 2020 Abstract We have reported sulfanilic acid as an exceedingly competent catalyst for one-pot Mannich reaction to give β- amino carbonyl compounds in good to excellent yield within a short reaction time. The various organic acids have screened; like gallic acid, 4-hydroxy benzoic acid, 4-amino benzoic acid, phenylacetic acid, chloroacetic acid, sulfosalicylic acid, sulfanilic acid, chloro benzoic acid, phthalic acid, salicylic acid, cinnamic acid, hippuric acid, 1- naphthyl acetic acid, o-amino benzoic acid, p-TSA, succinic acid, malic acid, and among them sulfanilic acid is a suitable catalyst. The reaction condition was optimized with respect to the solvent, the amount of catalyst as well as the variation of the ketone, aldehyde, and amine substrates. The procedure is mild, effective, ecofriendly, and the use of the minimum amount of catalyst. Keywords. Aldehydes, amino ketone, organic acid, multicomponent, Mannich reaction. 1. INTRODUCTION Multicomponent reaction is unique important reactions in organic synthesis. These reactions in the last two decades have acknowledged the consideration of organic chemists because of various merits over conventional traditional synthesis. Multicomponent reactions have significant applications in medicinal chemistry for the creation of diverse scaffolds and combinatorial libraries for drug development. [1] In the multicomponent reactions, three or more components have been reacted to form preferably one product, which has been containing the crucial units of all the original materials. MCRs have been good for the environment by decreasing the number of synthetic steps, energy consumption, and waste creation. Therefore, the discovery of novel MCRs and elaborating on the formerly known MCRs are substantial attention. Individual example of, this type is the preparation of β-amino carbonyl compounds by the Mannich reaction. Several β- amino ketones and their analogues, show the effective medicinal properties [2-13] are shown in figure 1. β-amino ketones, have vital intermediates in the preparation of various nitrogen-containing natural products, and pharmaceutically important compounds. [14] It is evidence that the reaction comprises two equilibrating constituents (imine formation and enol tautomerization) and therefore, demands harsh reaction conditions. Due to this, it is afflicted by some severe weaknesses, namely complex work-up, and purification procedures, and unwanted side products. [15] From the first report of the Mannich reaction, numerous studies have published to improve the reaction conditions and present new and efficient catalysts for this reaction. The Mannich reactions of ketones, aldehydes, and amines have been reported through Lewis acid, [16-19] Lewis base, [20] Brønsted acids, [21-23] Metal Triflates, [24-27] and transition metal salts. [28-30] Even with the merits of these methodologies; use of transition metal compounds and heavy metal compounds as the catalyst, preparation of the catalyst, problems recycling and reusing of the catalysts, toxic reagents, and solvents, drastic reaction conditions, toxicity, or difficulty in product separation has persisted disquiets. Hence, the exploration of the newfangled green method has stagnant being keenly pursued. On the other hand, because of growing concerns about environmental effects; execution of organic reactions using organocatalyst has extremely needed in recent years. Apart from being environmentally friendly, organocatalyst has obeyed green chemistry principle like casings mild conditions, consequently saving energy, oxygen-stable reagents and does not require anhydrous conditions, reducing the cost of the synthesis, less toxic and safer substances, stop the creation of metallic waste and avoids traces of Vietnam Journal of Chemistry Pramod Kulkarni © 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 676 metals in the products. [31] Here, we have used Sulfanilic acid as organocatalyst in a one-pot three- component Mannich reaction for the synthesis of β- amino ketone. Figure 1: Medicinal properties of β-amino ketone Sulfanilic acid is valuable, low-cost, and eco- friendly, and occurs as a grayish-white powder, which is stable and incompatible with strong oxidizing agents. The literature survey reveals that it has been applied as a catalyst for the synthesis of a variety of heterocyclic compounds. [32-39] The sulfanilic acid that occurs in solid form is present as a zwitterion. Sulfonic acid groups (-SO3H) can be deprotonated to become negative sulfonate (-SO3 - ) and amino groups which can be protonated to become positive ammonium groups (-NH3 + ). The zwitterionic property of sulfanilic acid may be responsible for the excellent catalytic property in organic synthesis. Sulfanilic acid is commercially available, economical, non polluting and easy to handle, non-inflammable, and a stable catalyst related to other organoacid catalysts which are eroding, costly and afford intensely acidic conditions. [40] Here we reported the use of sulfanilic as organocatalyst in one-pot three-component Mannich reaction for the synthesis of β-amino ketone. 2. MATERIALS AND METHODS All the starting materials were got from commercially accessible sources and used without further purifi ... ituted aryl aldehydes have given the corresponding product, in moderate yield, due to its higher crowded steric effects. Scheme 4: Sulfanilic acid-catalyzed one-pot three-component Mannich reaction between cyclohexanone, substituted benzaldehyde and aniline Table 4: Sulfanilic acid catalyzed Mannich reactions of ketones, various aldehydes and aniline a Entry Aldehyde Product Time (min.) % Yield b p.p. ( o C) 1 4 a 15 93 141-142 [41] 2 4b 25 90 113-115 [42] 3 4c 65 45 127-129 4 4 d 81 78 145-147 [41] 5 4e 84 125 157 Vietnam Journal of Chemistry Sulfanilic acid catalysed one-pot three-component © 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 683 6 4f 60 89 136-137 [43] 7 4g 35 82 127-128 8 4 h 10 88 154-155 [42] 9 4i 15 76 125-177 10 4j 45 80 171-172 a : Reaction condition: 5 mmol of Cyclohexanone, 5 mmol of aldehyde, 5 mmol of aniline and 20 mol% of sulfanilic acid in 10 ml ethanol under stirring condition at RT. b : All yields of isolated product. Next, we have extended the scope of the reaction with an aromatic ketone, various aromatic aldehydes, and various aromatic amine (scheme 5). The reaction has well proceeded with electron- donating and electron-withdrawing groups on aromatic aldehydes (table 5). The ortho-substituted benzaldehydes have been afforded a low yield due to steric hindrance. The 2-nitrobenzaldehyde did not give product due to, –M, -I, and steric effect. The reaction has proceeded well with aromatic amines, with electron-donating as well as an electron- withdrawing group. The ortho-substituted anilines with an electron-donating group have been afforded the low yield due to steric hindrance, while the electron-withdrawing group has not been afforded the product due to –M, -I, and steric effect. Scheme 5: Sulfanilic acid - catalyzed one-pot three-component Mannich reaction between acetophenone, substituted benzaldehyde and substituted aniline Vietnam Journal of Chemistry Pramod Kulkarni © 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 684 Table 5: Sulfanilic acid catalyzed Mannich reactions of acetophenone, aldehydes and substituted anilines a Entry Aldehyde Amines Product (8) Time (min.) Yield b m.p. (ºC) 1 8a 2 76 212 2 8b 600 NR c - 3 8c 400 82 164-166 [44] 4 8d 30 85 161-162 [45] 5 8e 240 78 113-114 [46] 6 8f 90 82 137-139 7 8g 45 70 118 8 8h 43 73 108 9 8i 500 40 145-147 Vietnam Journal of Chemistry Sulfanilic acid catalysed one-pot three-component © 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 685 10 8j 500 33 140-142 11 8k 45 24 109-111 12 8l 3 82 126-128 13 8m 125 78 134-137 14 8n 2 62 181-184 15 8o 200 60 164-166 16 8p 230 74 169-172 17 8q 400 NR c -- 18 8r 360 NR c - a : Reaction condition: 5 mmol of acetophenone, 5 mmol of substituted aldehyde, 5 mmol of substituted aniline and 20 mol% of catalyst in 10 ml ethanol under stirring condition at RT. b : All yields of isolated product. c NR: no reaction. Vietnam Journal of Chemistry Pramod Kulkarni © 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 686 4. CONCLUSION Here we report, an efficient synthesis of a series of β-amino carbonyl compounds from various ketones, aldehydes, and anilines using sulfanilic acid as an organocatalyst. The method has been congenial with various functional groups present on the aryl aldehydes, and anilines. The better yield of the product was perceived in the solvent than solvent- free conditions, and ethanol was found to be a best solvent for the reaction. The method has offered several merits, which include a small amount of catalyst loading, functional group tolerance, mild conditions, operational easiness, high yield, use of non-hazardous chemicals, and ecofriendly method. REFERENCES 1. A. Davoodnia, A. Tavakoli-Nishaburi, N. Tayakoli- Hoseini. Carbon-based solid acid catalyzed one-pot Mannich reaction: A facile synthesis of β-amino carbonyl compounds, Bull. Korean Chem. Soc., 2011, 32, 635-638. 2. G. X. Tang, J. F.Yan, L. Fan, J. Xu, X. L. Song, L. Jiang, L. F. Luo, D. C. Yang. Synthesis of novel β- amino ketones containing a p-aminobenzoic acid moiety and evaluation of their antidiabetic activities, Science China Chemistry, 2013, 56, 490-504. 3. H. I. Gul, A. A. Denizci, E. Erciyas. Antimicrobial evaluation of some Mannich bases of acetophenones and representative quaternary derivatives, Arzneimittelforschung, 2002, 52, 773-777. 4. H. I. Gul, U. Calis, J. Vepsalainen. Synthesis of some mono-Mannich bases and corresponding azines derivatives and evaluation of their anticonvulsant activity, Arzneimittelforschung, 2004, 54, 359-364. 5. H. I. Gul, A. Demirtas, G. Ucar, P. Taslimi, I. Gulcin. Synthesis of Mannich bases by two different methods and evaluation of their acetylcholine esterase and carbonic anhydrase inhibitory activities, Lett. Drug Design & Discovery, 2017, 14, 573-580. 6. J. R Dimmock, K. K. Sidhu, M. Chen, R. S. Reid, T. M. Allen, G. A. Truitt. Evaluation of some Mannich bases of cycloalkanones and related compounds for cytotoxic activity, Eur. J. Med. Chem., 1993, 28, 313-322. 7. H. I. Gul, T. Ojanen, O. Hanninen. Antifungal evaluation of bis Mannich bases derived from acetophenones and their corresponding piperidinols, Biol. Pharm. Bull., 2002, 25, 1307-1310. 8. K. K. Sivakumar, A. Rajasekaran, P. Senthilkumar, P. P. Wattamwar. Conventional and microwave assisted synthesis of pyrazolone Mannich bases possessing anti-inflammatory, analgesic, ulcerogenic effect and antimicrobial properties, Bioorg. Med. Chem. Lett., 2014, 24, 2940-2944. 9. R. Kenchappa, Y. D. Bodke, S. K. Peethambar, S. Telkar, V. K. Bhovi. Synthesis of β-amino carbonyl derivatives of coumarin and benzofuran and evaluation of their biological activity, Med. Chem. Es., 2013, 22, 4787-4797. 10. A. Arnold, F. Zhul, A. Kosinskil, T. J. Mangano, V. Setola, B. L. Roth, R. K. Guy. Improvement of pharmacological properties of irreversible thyroid receptor coactivator binding inhibitors, J. Med. Chem., 2009, 52, 3892-3901. 11. M. L. Edwards, H. W. Ritter, D. M. Stemerick, K. T. Stewart. Mannich bases of 4-phenyl-3-buten-2-one. A new class of antiherpes agent, J. Med. Chem., 1983, 26, 431-436. 12. R. F. Maisey, G. Jones, A. R. Somerville, B. A. Whittle. Substituted 1,1-diphenyl-3-aminoprop-1- enes and 1,1-diphenyl-3-aminopropanes as potential antidepressant agents, J. Med. Chem., 1971, 14, 161- 164. 13. E. Mete, H. I. Gul, C. Kazaz. Synthesis of 1-aryl-3- phenethylamino-1-propanone hydrochlorides as possible potent cytotoxic agents, Molecules, 2007, 12, 2579-2588. 14. R. Müller, H. Goesmann, H. Waldmann. N, N- Phthaloylamino acids as chiral auxiliaries in asymmetric Mannich-type reactions, Angew. Chem. Int. Ed., 1999, 38, 184-187. 15. C-T. Chang, B-S. Liao, S-T. Liu. Mannich-type reactions in a colloidal solution formed by sodium tetrakis(3,5-trifluoromethylphenyl)borate as a catalyst in water, Tetrahedron Lett., 2006, 47, 9257- 9259. 16. S. S. Mansoor, K. Aswin, K. Logaiya, S. P. N. Sudhan. An efficient synthesis of β-amino ketone compounds through one-pot three-component Mannich-type reactions using bismuth nitrate as catalyst, J. Saudi Chem. Soc., 2015, 19, 379-386. 17. Y. Wu, X. Wang, Y. Luo, J. Wang, Y. Jian, H. Sun, G. Zhang, W. Zhang, Z. Gao. Solvent strategy for unleashing the Lewis acidity of titanocene dichloride for rapid Mannich reaction, RSC Adv., 2016, 6, 15298-15303. 18. Y. Zhang, J. Han, Z-J. Liu. Diaryliodonium salts as efficient Lewis acid catalysts for direct three component Mannich reactions, RSC Adv., 2015, 5, 28485-25488. 19. R. Qiu, X. Xu, L. Peng, N. Li, S. Yin. Strong Lewis acids of air-stable metallocene bis(perfluorooctanesulfonate)s as high-efficiency catalysts for carbonyl-group transformation reactions, Chem. Eur. J., 2012, 18, 6172-6182. 20. Q. Guo, J. C-G. hao, H. Arman. Base-catalyzed three-component direct Mannich reaction of enolizable ketones with high syn-selectivities, Tetrahedron Lett., 2012, 53, 4866-4869. 21. Akiyama, J. Takaya, H. Kagoshima. Brønsted acid- catalyzed Mannich-type reactions in aqueous media, Adv. Synth. Cataly., 2002, 344, 338-347. 22. T. Akiyama, K. Matsuda, K. Fuchibe. HCl-catalyzed stereoselective Mannich reaction in H2O-SDS system, Synlett, 2005, 322-324. 23. M. Barbero, S. Cadamuro, S. Dughera. Brønsted acid Vietnam Journal of Chemistry Sulfanilic acid catalysed one-pot three-component © 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 687 catalyzed enantio- and diastereoselective one-pot three component Mannich reaction, Tetrahedron: Asymmetry, 2015, 26, 1180-1188. 24. S-B. Han, J-Y. Wei, X-C. Peng, R. Liu, S-S. Gong, Q. Sun. Hf(OTf)4 as a highly potent catalyst for the synthesis of Mannich bases under solvent-free conditions, Molecules, 2020, 25, 388. DOI: https://doi.org/10.3390/molecules25020388. 25. T. Ollevier, E. Nadeau. Bismuth Triflate-catalyzed three-component Mannich-type reaction, J. Org. Chem., 2004, 69, 9292-9295. 26. S. Yamasaki, T. Iida, M. Shibasaki. Direct catalytic asymmetric Mannich reaction of unmodified ketones: cooperative catalysis of an AlLi bis(binaphthoxide) complex and La(OTf)3.nH2O, Tetrahedron, 1999, 55, 8857-8867. 27. Y-Y. Yang, W-G. Shou, Y-G. Wang. Synthesis of β- amino carbonyl compounds via a Zn(OTf)2-catalyzed cascade reaction of anilines with aromatic aldehydes and carbonyl compounds, Tetrahedron, 2006, 62, 10079-10086. 28. S. Pal, L. H. Choudhury, T. Arvin. VCl3 catalyzed imine-based multicomponent reactions for the facile access of functionalized tetrahydropyridines and β- amino carbonyls, Mol. Diver., 2012, 16, 129-143. 29. R. Wang, B. Li, T. Huang, L. Shi, X. Lu. NbCl5 - catalyzed one-pot Mannich-type reaction: three component synthesis of β-amino carbonyl compounds, Tetrahedron Lett., 2007, 48, 2071-2073. 30. B. Eftekhari-Sis, A. Abdollahifar, M. M. Hashemi, M. Zirak. Stereoselective synthesis of β-amino ketones via direct Mannich-type reactions, catalyzed with ZrOCl2.8H2O under solvent-free conditions, Eur. J. Org., 2006, 5152-5157. 31. V. G. Oliveira, M. F. C. Cardoso, L. S. M. Forezi. Organocatalysis: A brief overview on its evolution and applications, Catalysts, 2018, 8, 605. 32. H. Kiyani, A. Mosallanezhad. Sulfanilic acid- catalyzed synthesis of 4-arylidene-3-substituted isoxazole-5(4H)-ones, Current Org. Synth., 2018, 15, 715-722. 33. R. M. Borik. Comparison on microwave and ultrasound accelerated synthetic route to dihydropyrimidinones catalyzed by sulfanilic acid in water, Aus. J. Basic and Applied Sci., 2013, 7, 543- 547. 34. U. P. Tarpada, B. B. Thummar, D. K. Raval. A green protocol for the synthesis of quinoxaline derivatives catalyzed by polymer supported sulphanilic acid, Arabian J. Chem., 2017, 10, S2902-S2907. 35. J. N. Sangsheeti, N. D. Kokare, D. B. Shinde. Sulfanilic acid catalyzed solvent-free synthesis of 1,5-benzodiazepine derivatives, Chinese Chem. Lett., 2007, 18, 1305-1308. 36. H. M. Mirzaei, B. Karimi. Sulfanilic acid as a recyclable bifunctional organocatalyst in the selective conversion of lignocellulosic biomass to 5-HMF, Green Chem., 2016, 18, 2282-2286. 37. B. Kumar, N. S. Rathore, K. L. Ameta. Synthesis of some new substituted oxiranes from 4’-hydroxy-3’5’- dinitrochalcones and their sulfanilic acid-catalyzed aminolysis, Res. Chem. Intermed., 2014, 40, 555-567. 38. A. F. Mohammed, N. D. Kokare, J. N. Sangshetti, D. B. Shinde. Sulfanilic acid catalyzed facile one-pot synthesis of 2,4,5-triarylimidazoles from benzyl/benzoin and aromatic aldehydes, J. Korean Chem. Soc., 2007, 51, 418-422. 39. F. Adam, K. M. Hello, T. H. Ali. Solvent free liquid- phase alkylation of phenol over solid sulfanilic acid catalyst, Applied Catalysis A: General, 2011, 399, 42-49. 40. R. K. Singh, B. Singh, R. Duvedi, S. Kumar. Sulfanilic acid: a versatile and efficient catalyst amongst various organoacids screened for the synthesis of 1-amido-2-naphthols under solvent-free condition, Res. Chem. Intermed., 2015, 41, 4083- 4099. 41. Q. Xu, Z. Yang, D. Yin, J. Wang. One-pot three- component Mannich reaction catalyzed by sucrose char sulfonic acid, Front. Chem. Eng. China, 2009, 3, 201-205. 42. J. Li-li, L. Yun-tao. One-pot three-component Mannich reaction catalyzed by 2-hydroxylpyridine, Chem. Res. Chin. Univ., 2013, 29, 710-713. 43. M. Kidwai, A. Jahan. Cerium chloride as a highly efficient catalyst for one-pot three-component Mannich reaction, J. Braz. Chem. Soc., 2010, 21, 21125-2179. 44. V. Yekkirala, R. K. Sudhagani, L. Panuganti. Yttrium(III) chloride catalyzed Mannich reaction: An efficient procedure for the synthesis of β-amino carbonyl compounds, Org. Commun., 2014, 7, 123- 129. 45. H. Li, H. Zeng, H. Shao. Bismuth(III) chloride- catalyzed one-pot Mannich reaction: three component synthesis of β-amino carbonyl compounds, Tetrahedron Lett., 2009, 50, 6858-6860. 46. B. Zhao, L. Jiang, Z. Liu, D. Qigang, L. Wang, B. Song, Y. Gao. Microwave-assisted Michael addition of 2-amino pyridine to chalcones under catalyst-free conditions, Res. Chem. Intermed., 2015, 49, 5809- 5819. Corresponding author: Pramod Kulkarni Department of Chemistry, Hutatma Rajguru Mahavidyalaya Rajgurunagar, Pune-410505, MS India E-mail: pramodskulkarni3@gmail.com.
File đính kèm:
 sulfanilic_acid_catalysed_one_pot_three_component_mannich_re.pdf
sulfanilic_acid_catalysed_one_pot_three_component_mannich_re.pdf

