The absorption properties of gold nano conjugated with proteins
The absorption properties of protein-conjugated metallic nanoparticles are
theoretically investigated based on the Mie theory and the core-shell model. Our
numerical calculations show that this finding is in good agreement with previous
experiments. We provide a better interpretation of the origin of optical peaks in the
absorption spectrum of the nanoparticle complex system. Our results can be used in
biomedical applications.

Trang 1

Trang 2

Trang 3

Trang 4
Trang 5

Trang 6

Trang 7
Bạn đang xem tài liệu "The absorption properties of gold nano conjugated with proteins", để tải tài liệu gốc về máy hãy click vào nút Download ở trên
Tóm tắt nội dung tài liệu: The absorption properties of gold nano conjugated with proteins

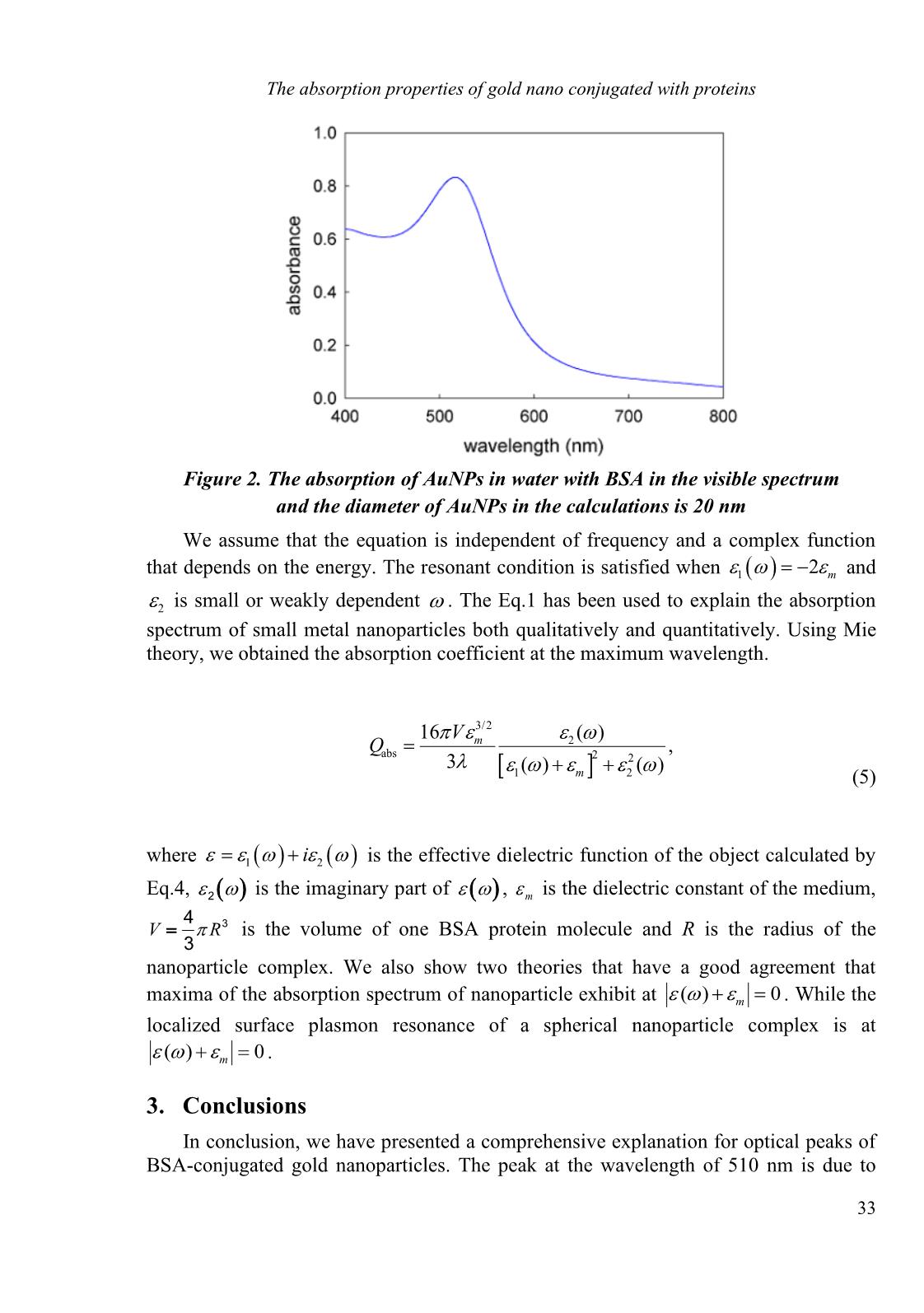
29 HNUE JOURNAL OF SCIENCE DOI: 10.18173/2354-1059.2020-0045 Natural Sciences 2020, Volume 65, Issue 10, pp. 29-35 This paper is available online at THE ABSORPTION PROPERTIES OF GOLD NANO CONJUGATED WITH PROTEINS Luong Thi Theu1, Le Anh Thi2, Tran Quang Huy1, Nguyen Quang Hoc3 and Nguyen Minh Hoa4 1Faculty of Physics, Hanoi Pedagogical University 2 2Institute of Research and Development, Duy Tan University 2Faculty of Natural Sciences, Duy Tan University 3Faculty of Physics, Hanoi National University of Education 4Faculty of Basic Sciences, Hue University of Medicine and Pharmacy Abstract. The absorption properties of protein-conjugated metallic nanoparticles are theoretically investigated based on the Mie theory and the core-shell model. Our numerical calculations show that this finding is in good agreement with previous experiments. We provide a better interpretation of the origin of optical peaks in the absorption spectrum of the nanoparticle complex system. Our results can be used in biomedical applications. Keywords: gold nanoparticle, BSA protein, Mie theory. 1. Introduction Gold nanoparticles (GNPs), with a diameter between 1 nm and 100 nm, have been widely used in chemical and biological sensors because of their excellent physical and chemical properties. The unique optical property of GNPs is one of the reasons that GNPs attract immense benefits from various fields of science, especially in the development of sensors. The spherical GNP solutions show a range of vibrant colors including red, blue, and violet when the particle size increases, and they can be used to dye glass in ancient times. The strong color is caused by the strong absorption and scattering of 520 nm light [1], which is the result of the collective oscillation of conduction electrons on the surface of GNPs when they are excited by the incident light. This phenomenon is called surface plasmon resonance (SPR), and it depends greatly on particle size and shape. Therefore, the SPR peak can be adjusted by manipulating the size of GNPs, and this property cannot be observed on bulk gold and GNPs with a diameter smaller than 2 nm. The SPR peak is not only sensitive to the size and the shape, but also many factors such as a protective ligand, refractive index of solvent, and temperature. The distance Received June 17, 2020. Revised October 16, 2020. Accepted October 23, 2020. Contact Nguyen Minh Hoa, e-mail address: [email protected] Luong Thi Theu, Le Anh Thi, Tran Quang Huy, Nguyen Quang Hoc and Nguyen Minh Hoa 30 between particles particularly shows the great influence on SPR. Thus, the red-shifting and the broadening of the peak are observed when GNPs are synthesized due to analyte binding. The color change of synthesized GNPs from red to blue is the principle of colorimetric sensors. Several recent pieces of research and reviews provide a detailed discussion of the factors that affect the SPR of GNPs [2-6]. Bovine serum albumin (BSA) protein has been widely used in the field of biophysics and medical science, due to its low cost, structural/ functional similarity to human serum albumin (HSA) [7]. A recent study found that the ribosylation of BSA resulted in reactive oxygen species (ROS) accumulation which killed breast cancer cells [8]. Particularly the anomalous thermal denaturing of proteins increased signal in the tests, biochemical reactions [9, 10]. This effect is strong in BSA proteins and is particularly useful for the design of bio-sensors and devices. In recent years, the plasmonic properties of metallic nanoparticles are of great interest because they have various potentially technological applications, especially the magnetic nanoparticles (NPs). Localized surface plasmon resonances with gold nanoparticles have many applications for a variety of application areas e.g. chemical analysis and catalytic, detect biomolecules, pharmaceutical, diagnosis, imaging, and therapy [1, 11, 12]. Complex systems of biological gold nanoparticles have also been investigated to construct functional devices for cell imaging, drug delivery, and biomolecule detection. Bovine Serum Albumin (BSA) proteins have been particularly useful in this issue [1]. The BSA substances not only prevent AuNPs from together combination but also are effective for treatment delivery and attaching AuNPs in living matter. Because of their large scattering crossing sections, BSA-AuNPs themselves can be imaged under white light illumination. Moreover, adjusting the optical plasmon resonance on the visible spectrum is implemented by changing the particle size and shape that have been especially helpful in optimizing the application of complex systems of biological gold nanoparticles. In this paper, we theoretically study the optical properties of AuBSA core-shell nano using the Mie theory and effective medium approximation, which has been synthesized experimentally in Ref. 13 in the visible range. 2. Content 2.1. Theoretical background Calculating exactly the number of BSA molecules on a gold nanoparticle’s surface based on the absorption spectrum and the extended Mie theory [13] shows that the core- shell model and the effective medium approximation provides a good agreement between theoretical calculations and experimental for spherical nanoparticles. Now, we apply these theories to the complex system to investigate and predict the properties of protein- conjugated gold nanoparticles. An idea of modeling nanoparticles conjugated nanoparticles as a core-shell structure has been widely used [14, 15]. In this work, the absorption and scattering of AuNP conjugated BSA protein in aqueous solutions are theoretically considered. The system is formed when BSA and AuNP proteins are placed in water. Some of the water is mixed with protein and this aqueous solution of BSA is attracted to AuNP through Van der Waals interaction. As a result, a protein conjugated nanoparticle is formed in water as shown in Figure 1. The absorption properties of gold nano conjugated with proteins 31 Figure 1. The core-shell model for protein-conjugated gold nano The general solution to the problem of scattering of a spherical metal sphere according to electrodynamics theory was first proposed by Mie in 1908 [16]. Mie's theory applied an overview theory of scattering on small particles to explain the color changing of the colloidal gold nanoparticles with arbitrary size and shows s good agreement with experimental results. When the radius of the nanoparticles is much smaller than the wavelength of the incident light ( d , or an approximation max /10d ), the Mie coefficients can be simplified by quasi-static approximations. Thus, using the exact solution of Mie theory is necessary to calculate accurately the extinction, scattering, and absorption coefficients cross-section of isotropically coated spherical nanoparticles are given by [17]. ( ) ( ) 1 2 2 scat a 1 sc t . 2 (2 1) Re , 2 (2 1) , ext ab n s ext n n n n n C n a b C n a C C b C = = = − = + + = + + (1) where ' ' ' ' ' ' ' ' 2 ( ) ( ) ( ) ( ) , ( ) ( ) ( ) ( ) ( ) ( ) ( ) ( ) , ( ) ( ) ( ) , ) ( p p p p n p p p p p p p p n p p p i i p m mka ka ka mka a m mka ka ka mka mka ka m ka mka b mka ka m ka ka C Q m R − = − − = = − (2) in which, Qi are the extinction, scattering, and absorption efficient, with i = [ext, scat, abs] is running index and R gold nanoparticle radius. is Riccati–Bessel function of the first and second kind, and s N m N = , Ns and N are the refractive index of the noble metal sphere NP r1 r2 Protein NP r1 Luong Thi Theu, Le Anh Thi, Tran Quang Huy, Nguyen Quang Hoc and Nguyen Minh Hoa 32 and the surrounding medium (perovskite), respectively, and 2 /k = is the wavenumber with the dielectric function of core-shell spherical and n represent the mode. Expansion of infinite series exhibits the different excitation symmetry like a dipole, quadrupole, and octupole corresponding to different values of n, respectively. Since it is very difficult to find the solution of the sum of infinite series, we can easily handle the situation if we truncated the series up to a certain value of n. If we are interested to study dipolar effects, choose n = 1, for quadrupolar n = 2, and so on. 2.2. The absorption efficiency of BSA protein-conjugated gold nano An effective dielectric function of core-shell nanoparticle dispersed in a solution can be found from Maxwell-Garnett theory as 2 2 1 1 1 2 2 2 2 2 1 1 1 2 2 2 2 1 2 2 1 , 1 2 , , a b a b r r r r r r r r = + + − = − + + = (3) in which, , 1 and 2 are the wavelength of the incident in a vacuum, the dielectric function for the core (Au), shell (BSA + surrounding medium), respectively. Parameters and analytical expressions for these dielectric functions can be taken from a previous study [18]. We introduce the filling factor of protein BSA on metallic surface f, 2 protein (1 ) mf f = + − , where m is the dielectric constant of the medium. na and nb are given by ( ) 2 2 2 2 1 ( ) ( ) , 2 8 1 ( ) log ( ) ( ) 2 4 1 2 ( ) ( ) . 2 4 m n m m m m m n m m R a kR i kR kR kR R b kR − = − + + − − − = + − + (4) Figure 2 shows the absorption efficiency of BSA conjugated gold nanoparticles in water. We found that Qabs behaves as a function of the wavelength. Here, we take that 1 10r = nm for the AuNP and 2 11.5r = nm for the shell. The recent experiments indicate that such a configuration corresponds to a BSA monolayer around the Au core [18]. There is a very good agreement with the reported data in Ref. 18 for the AuNP/water system. The absorption properties of gold nano conjugated with proteins 33 Figure 2. The absorption of AuNPs in water with BSA in the visible spectrum and the diameter of AuNPs in the calculations is 20 nm We assume that the equation is independent of frequency and a complex function that depends on the energy. The resonant condition is satisfied when ( )1 2 m = − and 2 is small or weakly dependent . The Eq.1 has been used to explain the absorption spectrum of small metal nanoparticles both qualitatively and quantitatively. Using Mie theory, we obtained the absorption coefficient at the maximum wavelength. 3/2 2 abs 2 2 1 2 16 ( ) , 3 ( ) ( ) m m V Q = + + (5) where ( ) ( )1 2i = + is the effective dielectric function of the object calculated by Eq.4, ( ) 2 is the imaginary part of ( ) , m is the dielectric constant of the medium, V R = 3 4 3 is the volume of one BSA protein molecule and R is the radius of the nanoparticle complex. We also show two theories that have a good agreement that maxima of the absorption spectrum of nanoparticle exhibit at ( ) 0m + = . While the localized surface plasmon resonance of a spherical nanoparticle complex is at ( ) 0m + = . 3. Conclusions In conclusion, we have presented a comprehensive explanation for optical peaks of BSA-conjugated gold nanoparticles. The peak at the wavelength of 510 nm is due to Luong Thi Theu, Le Anh Thi, Tran Quang Huy, Nguyen Quang Hoc and Nguyen Minh Hoa 34 biological molecules binding on nanoparticles and strongly depends on the dielectric function of the protein and the adsorption of protein on gold nanoparticles. The results show that there is a good agreement between theory and experiment. Our work shows that the finite size of the nanoparticles may play an important role in the plasmon spectral shift and it is directly related to the number of protein molecules attached to the AuNP surface. Acknowledgment. This work was financially supported by the Hue University of Science and Technology under grant number DHH2018-04-83. REFERENCES [1] Jain P.K., Lee K.S., El-Sayed I.H. et al., 2006. Calculated absorption and scattering properties of gold nanoparticles of different size, shape, and composition: Applications in biological imaging and biomedicine. J. Phys. Chem. B, 110(14), pp. 7238-7248. [2] Saha K., Agasti S.S., Kim C. et al., 2012. Gold nanoparticles in chemical and biological sensing. Chem. Rev., 112(5), pp. 2739-2779. [3] Trügler A., Tinguely J.C., Krenn J.R. et al., 2011. Influence of surface roughness on the optical properties of plasmonic nanoparticles. Phys. Rev. B - Condens Matter Mater Phys., 83(8). [4] Zeng S., Yong K.T., Roy I. et al., 2011. A Review on Functionalized Gold Nanoparticles for Biosensing Applications. Plasmonics, 6(3), pp. 491-506. [5] Jans H. và Huo Q., 2012. Gold nanoparticle-enabled biological and chemical detection and analysis. Chem. Soc. Rev., 41(7), pp. 2849-2866. [6] Philip R., Chantharasupawong P., Qian H. et al., 2012. Evolution of nonlinear optical properties: From gold atomic clusters to plasmonic nanocrystals. Nano Lett, 12(9), pp. 4661-4667. [7] Alsamamra H., 2019. Biophysical Interaction of Propylthiouracil with Human and Bovine Serum Albumins Materials and Samples Preparation, p. 1-7. [8] Khan M.S., Dwivedi S., Priyadarshini M. et al., 2013. Ribosylation of bovine serum albumin induces ROS accumulation and cell death in cancer line (MCF-7). Eur. Biophys. J., 42(11-12), pp. 811-818. [9] Lohcharoenkal W., Wang L., Chen Y.C. et al., 2014. Protein nanoparticles as drug delivery carriers for cancer therapy. Biomed Res. Int. [10] Thi Theu L., Thi Nhan T., Minh Hoa N. et al., 2016. Studying protein properties using fourth-order Ginzburg–Landau formalism. J. Sci. Nat. Sci., 61(4), pp. 39-44. [11] Lai L.M.H., Goon I.Y., Chuah K. et al., 2012. The Biochemiresistor: An Ultrasensitive Biosensor for Small Organic Molecules. Angew Chemie, 124(26), pp. 6562-6565. [12] Santra S., Dutta D., Walter G.A. et al., 2005. Fluorescent nanoparticle probes for cancer imaging. Technol Cancer Res Treat, 4(6), pp. 593-602. The absorption properties of gold nano conjugated with proteins 35 [13] Housni A., Ahmed M., Liu S. et al., 2008. Monodisperse protein stabilized gold nanoparticles via a simple photochemical process. J. Phy.s Chem. C, 112(32), pp. 12282-12290. [14] Phan A.D., Hoang T.X., Nghiem T.H.L. et al., 2013. Surface plasmon resonances of protein-conjugated gold nanoparticles on graphitic substrates. Appl. Phys. Lett., 103(16). [15] Phan A.D., Do N.C., and Nga D.T., 2017. Thermal-induced stress of plasmonic magnetic nanocomposites. J. Phys. Soc., Japan, 86(8). [16] Mie G., 1908. Beiträge zur Optik trüber Medien, speziell kolloidaler Metallösungen. Ann. Phys., 330(3), pp. 377-445. [17] Lee J.Y., Tsai M.C., Chen P.C. et al., 2015. Thickness Effects on Light Absorption and Scattering for Nanoparticles in the Shape of Hollow Spheres. J. Phys. Chem. C, 119(46), pp. 257540-25760. [18] Muskens O., Christofilos D., Del Fatti N. et al., 2006. Optical response of a single noble metal nanoparticle. J. Opt. A Pure Appl. Opt, 8(4).
File đính kèm:
 the_absorption_properties_of_gold_nano_conjugated_with_prote.pdf
the_absorption_properties_of_gold_nano_conjugated_with_prote.pdf

