Essential oils of Tunisian Pinus radiata D. Don, chemical composition and study of their herbicidal activity
Little is known about the phytotoxic effects of essential oils from Pine species, GC and GC/MS analysis of Pinus
radiata D. Don essential oil obtained from needles, resulted in the identification of 49 components comprising 97.6 %
of the oil totality. The composition was l dominated by monoterpene hydrocarbons (86.4 %) with β-pinene (40.2 %),
limonene (25.5 %) and α-pinene (15.2 %) were the major compounds. On the herbicidal activity, the oil strongly
inhibited seed germination and seedling growth of all tested weeds in a dose dependent manner with the effect being
significantly more effective on dicots (Sinapis arvensis L. and Trifolium campestre Schreb) than monocots (Phalaris
canariensis L.)

Trang 1

Trang 2

Trang 3
Trang 4
Trang 5

Trang 6
Tóm tắt nội dung tài liệu: Essential oils of Tunisian Pinus radiata D. Don, chemical composition and study of their herbicidal activity

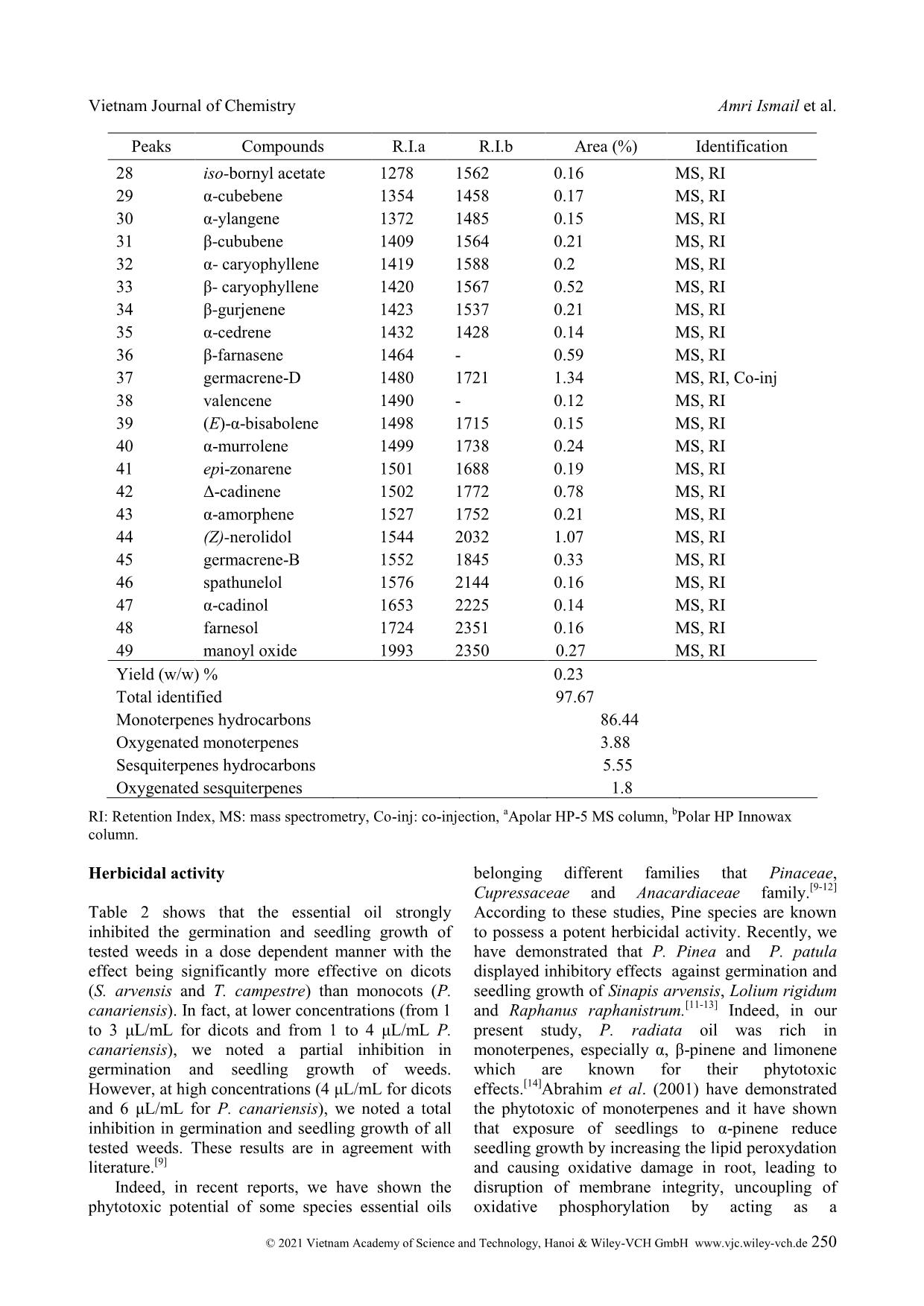
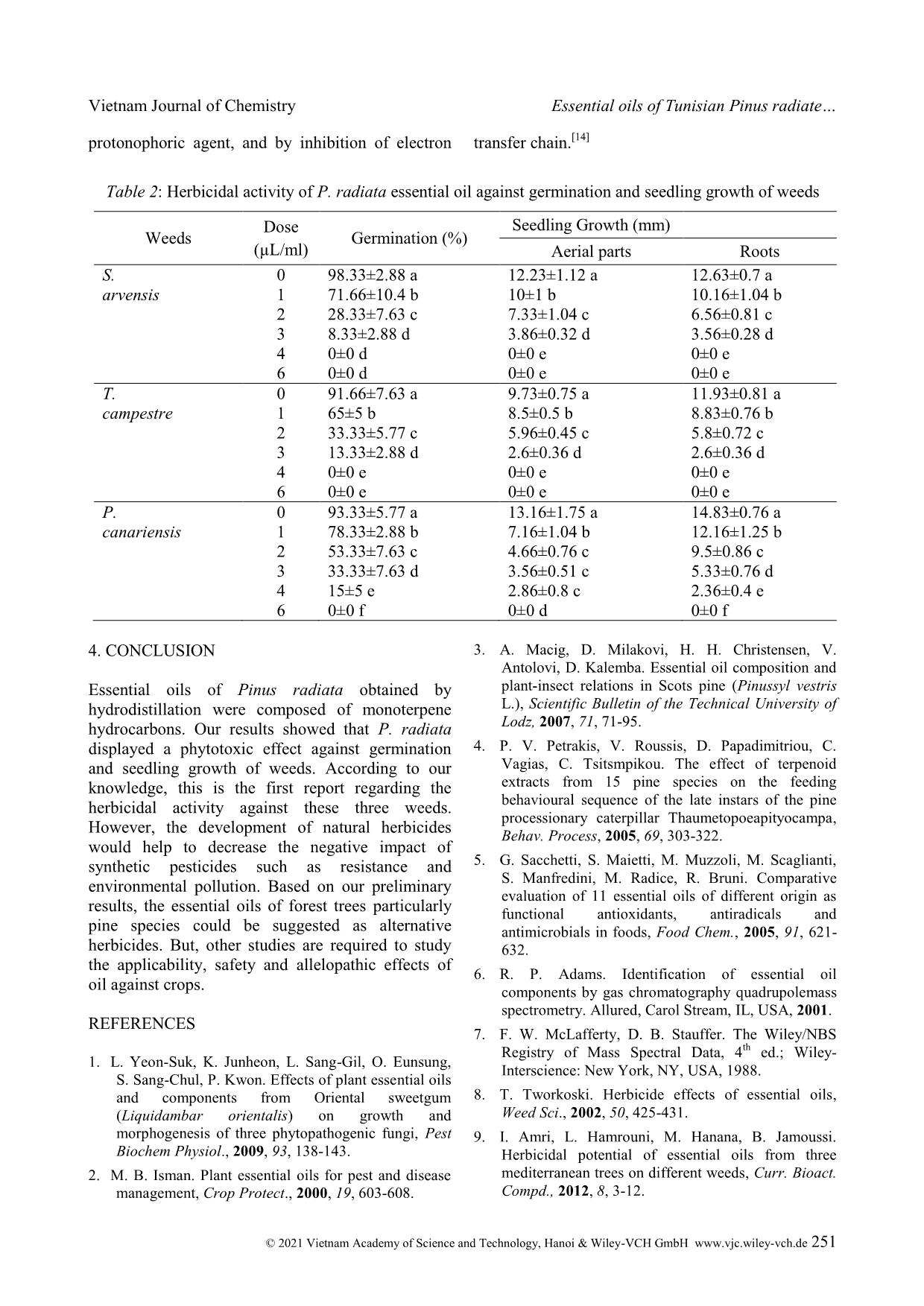
Cite this paper: Vietnam J. Chem., 2021, 59(2), 247-252 Article DOI: 10.1002/vjch.202000103 247 Wiley Online Library © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH Essential oils of Tunisian Pinus radiata D. Don, chemical composition and study of their herbicidal activity Amri Ismail¹ * , Kouki Habiba¹, Mabrouk Yassine¹, Hanana Mohsen 2 , Jamoussi Bassem 3 , Hamrouni Lamia 4 1 Laboratory of Biotechnology and Nuclear Technology, National Center of Nuclear Science and Technology (CNSTN), Sidi Thabet Technopark, Ariana, Tunisia 2 Plant Molecular Physiology Laboratory, Center of Biotechnology of Borj-Cedria, BP 901, 2050 Hammam-Lif, Tunisia 3 Chemistry Laboratory, Higher Institute of Education and Continuous Training, 43 Rue de la Libertelo 2019 Le Bardo, Tunisia 4 Laboratory for Forest Ecology, National Institute for Research in Rural Engineering, Water and Forests, BP 10, 2080 Ariana, Tunisia Submitted June 24, 2020; Accepted February 21, 2021 Abstract Little is known about the phytotoxic effects of essential oils from Pine species, GC and GC/MS analysis of Pinus radiata D. Don essential oil obtained from needles, resulted in the identification of 49 components comprising 97.6 % of the oil totality. The composition was l dominated by monoterpene hydrocarbons (86.4 %) with β-pinene (40.2 %), limonene (25.5 %) and α-pinene (15.2 %) were the major compounds. On the herbicidal activity, the oil strongly inhibited seed germination and seedling growth of all tested weeds in a dose dependent manner with the effect being significantly more effective on dicots (Sinapis arvensis L. and Trifolium campestre Schreb) than monocots (Phalaris canariensis L.). Keywords. Essential oils, Pinaceae, herbicidal activity, weeds. 1. INTRODUCTION In the last decades, scientists have been carried out to control plant diseases, particularly by the development of chemical pesticides. Although efficient, however their excessive applications in the crop lands and environment to avoid harmful pests resulted in an increased risk of enhanced pest resurgence and development of resistance, toxicological implications to non-target organisms and increased environmental pollution. [1] In fact, combating pollution and their harmful effects is a necessity for the intervention of scientists. For this, we must replace these synthetic pesticides with biological molecules, which are safer and do not induce any toxicological effects on the environment. The biological control of plant pest and diseases must be by using plant secondary metabolites, which play an important role in plant resistance to pests and several diseases. Therefore, screening plant essential oils and plant extracts for their biological activities could lead to discovery of new molecules for pest control. [2] Pinus radiata D. Don belongs to the Pinaceae family that comprises about 250 species which are divided into three subgenera, based on needles, seeds and cones characters: Ducampopinus, Strobus and Pinus. Pinus is the largest genus of conifers occurring naturally. [3] Essential oil composition of P. radiata has been previously studied by recent reports in Greece and Italy. [4,5] According to these reports, the major components of this oil were determined as β -pinene (16.8-35.21 %) and α-pinene (11.06-21.9 %). The antioxidant and radical-scavenging activities of P. radiata oil have been evaluated by means of 1,1- diphenyl-2-picrylhydrazyl assay, β-carotene bleaching test and luminol-photochemiluminescence assay. The antimicrobial properties of the oil were tested on five food-spoilage yeasts. [5] Moreover, knowing that production of essential oils depend on several factors that geographical origin and genetic background and to the best of our knowledge, the Vietnam Journal of Chemistry Amri Ismail et al. © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 248 chemical composition of Tunisian P. radiata essential oil has not been published and there is no report on the herbicidal effects of P. radiata oil, therefore the aims of this study are, in a first step, to study the chemical composition of the essential oil of P. radiata growing in Tunisia, and in a second step, we evaluated their herbicidal activity against the germination and seedling growth of weeds. 2. MATERIALS AND METHODS 2.1. Plant Material The needles of Pinus radiata D. Don were collected during month of October from the Souinet arboreta of the National Institute of Researches on Rural Engineering, Water and Forests. Five batches of needles were taken from different trees were harvested, and mixed to have a homogeneous sample. The experimental site is characterized by a humid climate at an altitude of 492 m. this site is located in Ain Draham, governorate of Jendouba, in the north of Tunisia. The plant was identified by Dr Lamia HAMROUNI, National Institute of Researches on Rural Engineering, Water and Forests, TUNISIA and a sample (PR-1109) was submitted to the herbarium division of the Institute. 2.2. Isolation of the essential oils Three replications of 100 g of air-dried and finely grounded needles were submitted to hydrodistillation for 3 hours with 500 ml distilled water using a Clevenger type apparatus. The volatile oils were collected and dried over anhydrous sodium sulfate and stored in sealed glass brown vials in a refrigerator at 4 °C. Yield based on dried weight of the sample was calculated (w/w %). 2.3. Gas chromatography and mass spectrometry analysis 2.3.1. Gas chromatography analysis with FID detection The essential oils were analyzed using a Hewlett Packard 5890 II GC equipped with Flame Ionization Detector (FID) and HP-5 MS capillary column (5 % phenyl/95 % dimethyl polysiloxane ... n hexane (1/100V/V). Volumes of 1μL were injected in the splitless mode. The relative percentage of each component was calculated electronically from FID area percent data. 2.3.2. Gas chromatography analysis with MS detection Analysis of P. radiata essential oil was carried out using a Hewlett Packard 5890 II GC, equipped with a capillary column HP-5 MS (30 m×0.25 mm, film thickness 0.25μm) and mass selective detector HP 5972. The oven temperature was kept at 50 °C for 1 min then programmed from 50 °C to 250 °C at 5 °C/min and subsequently, held isothermal for 4 min. The carrier gas was Helium at a flow rate of 1.2 mL/min. In GC/MS detection, an electron ionization system with a scan time of 1.5 s and mass range 40- 300 amu with ionization energy of 70 eV was used. Injector was set at 250 and transfer line temperatures at 280 °C. Samples diluted in hexane (1/10 V/V) of 1μl were injected in the splitless mode. The identification of oil components was based on mass spectra (compared with Wiley 275.L, 6th edition mass spectral library) or with standard compounds and experienced by comparing their retention index with those of authentic compounds or based on the results published in the literature. [6,7] Other confirmation was done from data generated from a series of n-alkanes retention indices (C9-C28) on HP-5 MS capillary column. 2.4. Seed germination and seedling growth experiments Mature seeds of weeds of Sinapis arvensis L., Phalaris canariensis L. and Trifolium campestre Schreb were collected from parent plants growing in fields in July. Seeds were sterilized with 15% sodium hypochlorite for 20 min. Then, they were rinsed with distilled water. Next, oils were dissolved in tween–water solution 1 % (V/V). The final concentrations of treatments were (0, 1, 2, 3, 4 and 6μL/mL). Solutions of 8 ml were transferred on the layers of filter paper placed in the Petri dish. Afterward, 20 seeds from each weed were placed on the filter paper. Petri dishes were closed with an adhesive tape and were incubated at 25 °C on a growth chamber equipped with 12 h of light. [8] The number of germinated seeds and seedling lengths were measured after 10 days and all tests were arranged in a completely randomized design with three replications by treatment. 2.5. Statistical analysis Vietnam Journal of Chemistry Essential oils of Tunisian Pinus radiate © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 249 Data of seed germination and seedling growth were subjected to one-way analysis of variance (ANOVA) using the SPSS 13.0 software package. Differences between means were evaluated by Student - Newman-Keuls test and means with values of P ≤ 0.05 were considered significantly different. 3. RESULTS AND DISCUSSION The hydro-distillation of dried Pinus radiata needles gave yellowish essential oil (yield 0.23 %, w/w). The chromatographic analysis showed a mixture of components belonging four subclass of compounds with a significant fraction of hydrocarbonated sesquiterpenes and monoterpenes. The list of the compounds, in order of their elution on the apolar HP-5 MS column, and the quantitative data, are reported in table 1. 49 oil compounds were identified accounting for 97.67 % of the total oil. Monoterpene hydrocarbons displayed the highest contribution (86.44 %) amongst which β-pinene (40.23 %), limonene (25.5 %) and α-pinene (15.22 %) were the most abundant. Whereas oxygenated monoterpenes were represented only by 3.88 %. In comparison with monoterpenes, sesquiterpenes were relatively weak (7.35 %); with 5.55 % of sesquiterpene hydrocarbons and oxygenated sesquiterpenes were the poorest fraction (1.8 %). The essential oil of P. radiate was previously investigated in Greece and Italy. The obtained results were in agreement with our data; it was shown that β-pinene (16.8-35.21 %) and α-pinene (11.06-35.21 %) were the major components. However, limonene is most abundant in oils from Tunisian P. radiate (25.5 %) than the sample of Greece (4.42 %) and Italy (12.6 %). [4,5] These differences could be related to the environmental factors (climate and soils), the genetic diversity and the extraction conditions. Table 1: Chemical composition of P. radiata essential oil Peaks Compounds R.I.a R.I.b Area (%) Identification 1 tricyclene 926 1014 0.1 MS, RI 2 α-thujene 931 1030 0.1 MS, RI 3 α-pinene 939 1033 15.22 MS, RI, Co-inj 4 α-fenchene 950 1059 0.1 MS, RI 5 camphene 950 1068 0.4 MS, RI, Co-inj 6 β-pinene 976 980 40.23 MS, RI 7 β-myrcene 991 1152 1.39 MS, RI, Co-inj 8 α-phellandrene 1007 1160 0.25 MS, RI 9 δ-3-carene 1011 1148 1.46 MS, RI 10 α-terpinene 1016 1183 0.1 MS, RI 11 p-cymene 1026 1258 0.1 MS, RI 12 limonene 1033 1032 25.5 MS, RI 13 (Z)-β-ocimene 1040 1230 0.77 MS, RI 14 δ-terpinene 1062 1236 0.1 MS, RI, Co-inj 15 α-terpinolene 1088 1280 0.62 MS, RI 16 linalool 1098 1547 0.34 MS, RI 17 α-fenchol 1098 1571 0.1 MS, RI 18 (Z)-pinocarveol 1141 - 0.34 MS, RI 19 camphor 1143 1473 0.11 MS, RI 20 δ-terpineol 1163 1662 0.88 MS, RI 21 carvone 1198 1636 0.1 MS, RI 22 pinocarvone 1164 - 0.11 MS, RI 23 myrtenol 1176 1586 0.43 MS, RI 24 terpen-4-ol 1179 1571 0.16 MS, RI 25 α-terpineol 1196 1673 0.88 MS, RI 26 verbenone 1204 1733 0.13 MS, RI 27 (Z)-carveol 1219 - 0.14 MS, RI Vietnam Journal of Chemistry Amri Ismail et al. © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 250 Peaks Compounds R.I.a R.I.b Area (%) Identification 28 iso-bornyl acetate 1278 1562 0.16 MS, RI 29 α-cubebene 1354 1458 0.17 MS, RI 30 α-ylangene 1372 1485 0.15 MS, RI 31 β-cububene 1409 1564 0.21 MS, RI 32 α- caryophyllene 1419 1588 0.2 MS, RI 33 β- caryophyllene 1420 1567 0.52 MS, RI 34 β-gurjenene 1423 1537 0.21 MS, RI 35 α-cedrene 1432 1428 0.14 MS, RI 36 β-farnasene 1464 - 0.59 MS, RI 37 germacrene-D 1480 1721 1.34 MS, RI, Co-inj 38 valencene 1490 - 0.12 MS, RI 39 (E)-α-bisabolene 1498 1715 0.15 MS, RI 40 α-murrolene 1499 1738 0.24 MS, RI 41 epi-zonarene 1501 1688 0.19 MS, RI 42 Δ-cadinene 1502 1772 0.78 MS, RI 43 α-amorphene 1527 1752 0.21 MS, RI 44 (Z)-nerolidol 1544 2032 1.07 MS, RI 45 germacrene-B 1552 1845 0.33 MS, RI 46 spathunelol 1576 2144 0.16 MS, RI 47 α-cadinol 1653 2225 0.14 MS, RI 48 farnesol 1724 2351 0.16 MS, RI 49 manoyl oxide 1993 2350 0.27 MS, RI Yield (w/w) % 0.23 Total identified 97.67 Monoterpenes hydrocarbons Oxygenated monoterpenes 86.44 3.88 Sesquiterpenes hydrocarbons 5.55 Oxygenated sesquiterpenes 1.8 RI: Retention Index, MS: mass spectrometry, Co-inj: co-injection, a Apolar HP-5 MS column, b Polar HP Innowax column. Herbicidal activity Table 2 shows that the essential oil strongly inhibited the germination and seedling growth of tested weeds in a dose dependent manner with the effect being significantly more effective on dicots (S. arvensis and T. campestre) than monocots (P. canariensis). In fact, at lower concentrations (from 1 to 3 μL/mL for dicots and from 1 to 4 μL/mL P. canariensis), we noted a partial inhibition in germination and seedling growth of weeds. However, at high concentrations (4 μL/mL for dicots and 6 μL/mL for P. canariensis), we noted a total inhibition in germination and seedling growth of all tested weeds. These results are in agreement with literature. [9] Indeed, in recent reports, we have shown the phytotoxic potential of some species essential oils belonging different families that Pinaceae, Cupressaceae and Anacardiaceae family. [9-12] According to these studies, Pine species are known to possess a potent herbicidal activity. Recently, we have demonstrated that P. Pinea and P. patula displayed inhibitory effects against germination and seedling growth of Sinapis arvensis, Lolium rigidum and Raphanus raphanistrum. [11-13] Indeed, in our present study, P. radiata oil was rich in monoterpenes, especially α, β-pinene and limonene which are known for their phytotoxic effects. [14] Abrahim et al. (2001) have demonstrated the phytotoxic of monoterpenes and it have shown that exposure of seedlings to α-pinene reduce seedling growth by increasing the lipid peroxydation and causing oxidative damage in root, leading to disruption of membrane integrity, uncoupling of oxidative phosphorylation by acting as a Vietnam Journal of Chemistry Essential oils of Tunisian Pinus radiate © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 251 protonophoric agent, and by inhibition of electron transfer chain. [14] Table 2: Herbicidal activity of P. radiata essential oil against germination and seedling growth of weeds Weeds Dose (µL/ml) Germination (%) Seedling Growth (mm) Aerial parts Roots S. 0 98.33±2.88 a 12.23±1.12 a 12.63±0.7 a arvensis 1 71.66±10.4 b 10±1 b 10.16±1.04 b 2 28.33±7.63 c 7.33±1.04 c 6.56±0.81 c 3 8.33±2.88 d 3.86±0.32 d 3.56±0.28 d 4 0±0 d 0±0 e 0±0 e 6 0±0 d 0±0 e 0±0 e T. 0 91.66±7.63 a 9.73±0.75 a 11.93±0.81 a campestre 1 65±5 b 8.5±0.5 b 8.83±0.76 b 2 33.33±5.77 c 5.96±0.45 c 5.8±0.72 c 3 13.33±2.88 d 2.6±0.36 d 2.6±0.36 d 4 0±0 e 0±0 e 0±0 e 6 0±0 e 0±0 e 0±0 e P. 0 93.33±5.77 a 13.16±1.75 a 14.83±0.76 a canariensis 1 78.33±2.88 b 7.16±1.04 b 12.16±1.25 b 2 53.33±7.63 c 4.66±0.76 c 9.5±0.86 c 3 33.33±7.63 d 3.56±0.51 c 5.33±0.76 d 4 15±5 e 2.86±0.8 c 2.36±0.4 e 6 0±0 f 0±0 d 0±0 f 4. CONCLUSION Essential oils of Pinus radiata obtained by hydrodistillation were composed of monoterpene hydrocarbons. Our results showed that P. radiata displayed a phytotoxic effect against germination and seedling growth of weeds. According to our knowledge, this is the first report regarding the herbicidal activity against these three weeds. However, the development of natural herbicides would help to decrease the negative impact of synthetic pesticides such as resistance and environmental pollution. Based on our preliminary results, the essential oils of forest trees particularly pine species could be suggested as alternative herbicides. But, other studies are required to study the applicability, safety and allelopathic effects of oil against crops. REFERENCES 1. L. Yeon-Suk, K. Junheon, L. Sang-Gil, O. Eunsung, S. Sang-Chul, P. Kwon. Effects of plant essential oils and components from Oriental sweetgum (Liquidambar orientalis) on growth and morphogenesis of three phytopathogenic fungi, Pest Biochem Physiol., 2009, 93, 138-143. 2. M. B. Isman. Plant essential oils for pest and disease management, Crop Protect., 2000, 19, 603-608. 3. A. Macig, D. Milakovi, H. H. Christensen, V. Antolovi, D. Kalemba. Essential oil composition and plant-insect relations in Scots pine (Pinussyl vestris L.), Scientific Bulletin of the Technical University of Lodz, 2007, 71, 71-95. 4. P. V. Petrakis, V. Roussis, D. Papadimitriou, C. Vagias, C. Tsitsmpikou. The effect of terpenoid extracts from 15 pine species on the feeding behavioural sequence of the late instars of the pine processionary caterpillar Thaumetopoeapityocampa, Behav. Process, 2005, 69, 303-322. 5. G. Sacchetti, S. Maietti, M. Muzzoli, M. Scaglianti, S. Manfredini, M. Radice, R. Bruni. Comparative evaluation of 11 essential oils of different origin as functional antioxidants, antiradicals and antimicrobials in foods, Food Chem., 2005, 91, 621- 632. 6. R. P. Adams. Identification of essential oil components by gas chromatography quadrupolemass spectrometry. Allured, Carol Stream, IL, USA, 2001. 7. F. W. McLafferty, D. B. Stauffer. The Wiley/NBS Registry of Mass Spectral Data, 4 th ed.; Wiley- Interscience: New York, NY, USA, 1988. 8. T. Tworkoski. Herbicide effects of essential oils, Weed Sci., 2002, 50, 425-431. 9. I. Amri, L. Hamrouni, M. Hanana, B. Jamoussi. Herbicidal potential of essential oils from three mediterranean trees on different weeds, Curr. Bioact. Compd., 2012, 8, 3-12. Vietnam Journal of Chemistry Amri Ismail et al. © 2021 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 252 10. I. Amri, L. Hamrouni, M. Hanana, B. Jamoussi. Chemical composition of Juniperus oxycedrus L. subsp macrocarpa essential oil and study of their herbicidal effects on germination and seedling growth of weeds, Asian J. Appl. Sci., 2011, 8, 771-779. 11. I. Amri, L. Hamrouni, S. Gargouri, M. Hanana, M. Mahfoudhi, T. Fezzani, E. Ferjani, B. Jamoussi. Chemical composition and biological activities of essential oils of Pinuspatula, Nat. Prod. Commun., 2011, 6, 1531-1536. 12. I. Amri, S. Gargouri, L. Hamrouni, M. Hanana, T. Fezzani, B. Jamoussi. Comparative study of two coniferous species (Pinus pinaster aiton and Cupressus sempervirens L. var. dupreziana [A. Camus] Silba) essential oils: Chemical composition and biological activity, Chil. J. Agric. Res., 2013, 73, 259-266. 13. A. Ben Ghnaya, L. Hamrouni, I. Amri, H. Ahoues, M. Hanana, A. Romane. Study of allelopathic effects of Eucalyptus erythrocorys L. crude extracts against germination and seedling growth of weeds and wheat, Nat. Prod. Res., 2016, 30, 2058-2064. 27. D. Abrahim, A. C. Francischini, E. M. Pergo, A. M. Kelmer-Bracht, E. Ishii-Iwamoto. Effects of α-pinene on the mitochondrial respiration of maize seedling, Plant Physiol. Biochem., 2003, 41, 985-991. Corresponding author: AMRI Ismail Laboratory of Biotechnology and Nuclear Technology National Center of Nuclear Science and Technology (CNSTN) Sidi Thabet Technopark, Ariana, Tunisia E-mail: amri_amri@live.fr Tel.: 0021652832495.
File đính kèm:
 essential_oils_of_tunisian_pinus_radiata_d_don_chemical_comp.pdf
essential_oils_of_tunisian_pinus_radiata_d_don_chemical_comp.pdf

