Tính toán độ dịch chuyển hóa học 13C của bakuchiol‒tác nhân mới điều trị tổn thương các cơ quan
The calculations of 13C NMR chemical shifts for bakuchiol, a promising anti-aging agent, were performed using 11
functionals (B3LYP, B3PW91, BPV86, CAM-B3LYP, HCTH, HSEH1PBE, mPW1PW91, PBEPBE, TPSSTPSS, and
ωB97XD) and 10 common basis sets (3-21G, 6-31G(d,p), 6-31G(d,3p), 6-31G(3d,p) 6-31G++(d,p), DGDZVP,
DGDZVP2, LANL2DZ, LANL2MB) to compare with experimental data. While functionals did not strongly impact the
computed 13C chemical shifts, basis sets showed a significant influence on the results. For those functionals, B3LYP,
B3PW91, CAM-B3LYP, HSEH1PBE, mPW1PW91, and ωB97XD were found to have strong correlations (r2 ≥ 0.9987)
and low errors (CMAEs ≤ 1.96 ppm and CMAEs ≤ 2.49 ppm); among the tested basis sets 3-21G, DGDZVP provided
the best results (r2 ≥ 0.9980, CMAEs ≤ 2.37 ppm and CMAEs ≤ 2.67 ppm). These results would allow meaningful
predictions of 13C chemical shifts for bakuchiol

Trang 1

Trang 2

Trang 3
Trang 4

Trang 5

Trang 6

Trang 7
Tóm tắt nội dung tài liệu: Tính toán độ dịch chuyển hóa học 13C của bakuchiol‒tác nhân mới điều trị tổn thương các cơ quan

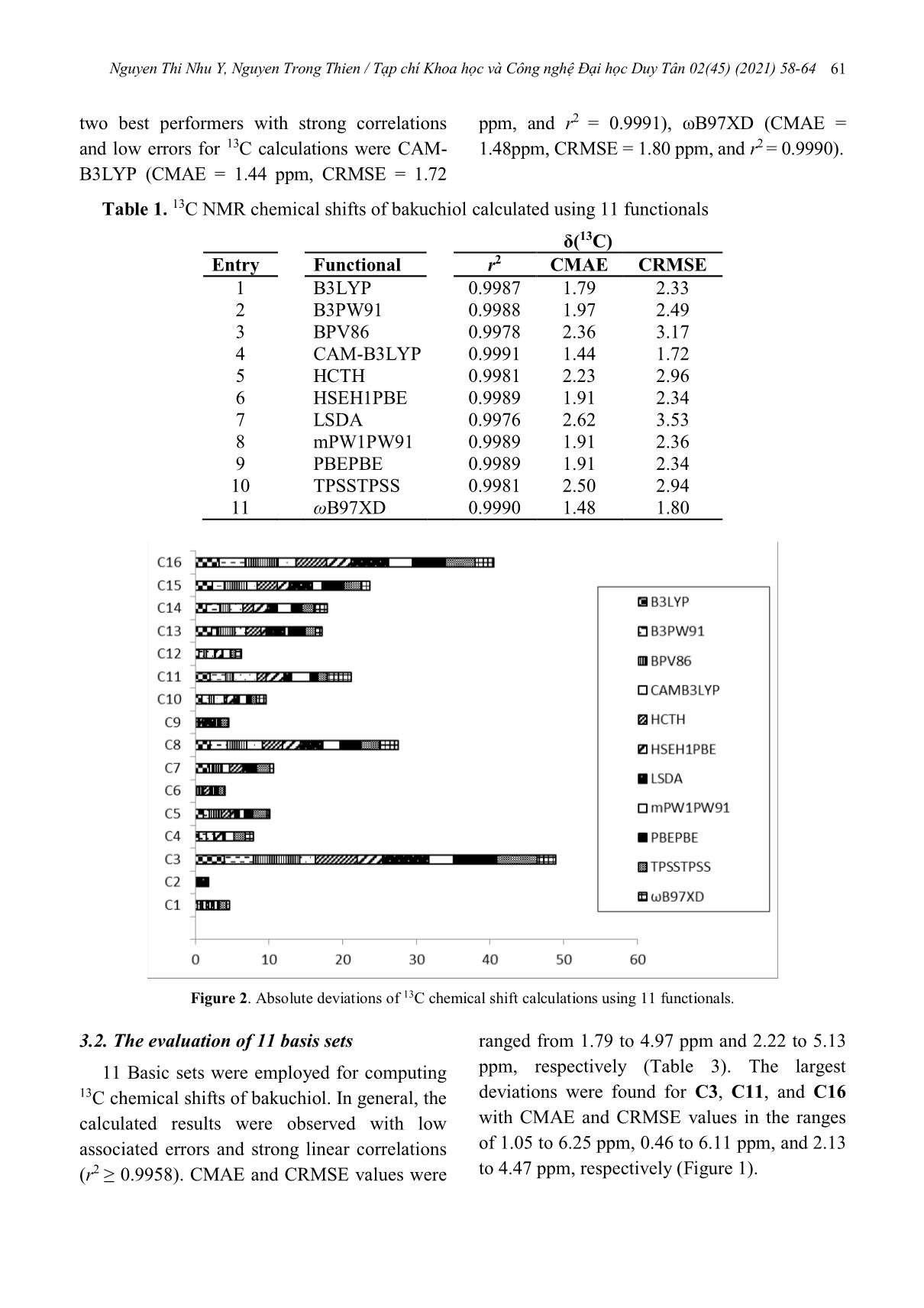
Nguyen Thi Nhu Y, Nguyen Trong Thien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 58-64 58 13C chemical shift predictions for bakuchiol‒a recently discovered agent against organ damage Tính toán độ dịch chuyển hóa học 13C của bakuchiol‒tác nhân mới điều trị tổn thương các cơ quan Nguyen Thi Nhu Ya, Nguyen Trong Thiena,b* Nguyễn Thị Như Ý a, Nguyễn Trọng Thiệna,b* aFaculty of Pharmacy, College of Medicine and Pharmacy Duy Tan University, Da Nang 550000, Vietnam bKhoa Dược, Trường Y- Dược, Đại học Duy Tân, Đà Nẵng, Việt Nam bInstitute of Research and Development, Duy Tan University, Da Nang 550000, Vietnam bViện Nghiên cứu và Phát triển Công nghệ Cao, Đại học Duy Tân, Đà Nẵng, Việt Nam (Ngày nhận bài: 03/12/2020, ngày phản biện xong: 07/01/2021, ngày chấp nhận đăng: 13/03/2021) Abstract The calculations of 13C NMR chemical shifts for bakuchiol, a promising anti-aging agent, were performed using 11 functionals (B3LYP, B3PW91, BPV86, CAM-B3LYP, HCTH, HSEH1PBE, mPW1PW91, PBEPBE, TPSSTPSS, and ωB97XD) and 10 common basis sets (3-21G, 6-31G(d,p), 6-31G(d,3p), 6-31G(3d,p) 6-31G++(d,p), DGDZVP, DGDZVP2, LANL2DZ, LANL2MB) to compare with experimental data. While functionals did not strongly impact the computed 13C chemical shifts, basis sets showed a significant influence on the results. For those functionals, B3LYP, B3PW91, CAM-B3LYP, HSEH1PBE, mPW1PW91, and ωB97XD were found to have strong correlations (r2 ≥ 0.9987) and low errors (CMAEs ≤ 1.96 ppm and CMAEs ≤ 2.49 ppm); among the tested basis sets 3-21G, DGDZVP provided the best results (r2 ≥ 0.9980, CMAEs ≤ 2.37 ppm and CMAEs ≤ 2.67 ppm). These results would allow meaningful predictions of 13C chemical shifts for bakuchiol. Keywords: 13C chemical shifts; NMR; DFT functionals; basis sets; bakuchiol. Tóm tắt Phổ 13C của bakuchiol, tác nhân chống lão hóa, được tính toán bằng 11 hàm mật độ (B3LYP, B3PW91, BPV86, CAM- B3LYP, HCTH, HSEH1PBE, mPW1PW91, PBEPBE, TPSSTPSS, và ωB97XD) và 10 mức lý thuyết (3-21G, 6- 31G(d,p), 6-31G(d,3p), 6-31G(3d,p) 6-31G++(d,p), DGDZVP, DGDZVP2, LANL2DZ, LANL2MB) nhằm so sánh với dữ liệu thực nghiệm. Trong khi các hàm mật độ thể hiện ảnh hưởng nhỏ lên độ dịch chuyển hóa học 13C, các kết quả tính toán bằng mức lý thuyết cho thấy sự phân hóa rộng hơn về độ chính xác. B3LYP, B3PW91, CAM-B3LYP, HSEH1PBE, mPW1PW91, và ωB97XD có độ tương quan cao (r2 ≥ 0.9987) và lỗi thấp (CMAEs ≤ 1.97 ppm và CMAEs ≤ 2.49 ppm); trong các mức lý thuyết, 3-21G, DGDZVP cho các kết quả với độ chính xác cao (r2 ≥ 0.9980, CMAEs ≤ 2.37 ppm and CMAEs ≤ 2.67 ppm). Từ khóa: Phổ 13C; NMR; hàm DFT; mức lý thuyết; bakuchiol. *Corresponding Author: Nguyen Trong Thien; Faculty of Pharmacy, College of Medicine and Pharmacy Duy Tan University, Da Nang 550000, Vietnam; Institute of Research and Development, Duy Tan University, Da Nang 550000, Vietnam Email: nguyentrongthien@duytan.edu.vn 02(45) (2021) 58-64 Nguyen Thi Nhu Y, Nguyen Trong Thien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 58-64 59 1. Introduction Bakuchiol (Figure 1), a prenylated phenolic monoterpene isolated from the fruit of Psoralea corylifolia, has recently shown a variety of pharmacological effects such as antioxidant, anti-bacterial, anti-inflammatory, anti-aging, and estrogen-like effects[1][2]. It also has protective effects in the heart, liver skin, and other organs. In addition, bakuchiol inhibits the proliferation of various cancer cells, including stomach, breast, and skin cancer cells and liverfibrosis via promoting myofibroblast apoptosis. It relieves the hepatotoxic of multiple toxicants by suppressing oxidative stress and inflammatory changes[3]. Understanding the structure of bakuchiol would provide insights into its pharmacological effects. Figure 1. (A) Bakuchiol and (B) its optimized structure at the IEFPCM(CHCl3)/B3LYP-631G(d,p) level of theory with numbered carbons (H atoms were omitted for clarity). The combination of experimental and computational NMR techniques has been a strong tool for providing the structural information of biologically active natural products, which can support the difficult assignments and the confirmation of their structures and provide valuable insights into the electronic environments of active NMR nucleus [4][5][6]. The gauge-including atomic orbitals (GIAO)/density functional theory (DFT) method are generally accepted as a standard method in computing shielding constants due to its reliability and applicability [7][8][9]. The accuracy of calculated chemical shifts typically depends on an appropriate combination of exchange-correlation functionals and basis sets [10]. Aimed to find suitable methods with high accuracy, this present study evaluated 11 DFT functionals and 11 common basis sets in the calculations of 13C chemical shifts for bakuchiol. 2. Computational methods All calculations were performed using the Gaussian09 [11]. Geometry optimizations of bakuchiol were performed at the IEFPCM(CHCl3)/B3LYP/6-31G(d,p) level[12][13]. Subsequent frequency calculations ensured that a potential energy surface (PES) local minimum was attained during the energy minimization. Cartesian coordinates of the resulting structures are given in the Supporting Information. The following 11 functionals coupled with 6-31G(d,p) [14] and 10 basis set coupled with B3LYP [15] were evaluated: - Funtionals: B3LYP (Becke’s 3-parameter hybrid functional[16] using B exchange[17] and LYP correlation),[15] B3PW91 (Perdew and Wang’s 1991 gradient-corrected correlation functional),[18][19] BPV86 (Perdew’s 1986 functional),[16][20][21] CAM-B3LYP (Handy Nguyen Thi Nhu Y, Nguyen Trong Thien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 58-64 60 and co-workers’ long-range corrected version of B3LYP using the Coulomb-attenuating ... viations. Overall, the correlation coefficients and error results indicate that the calculations provided a qualitatively accurate description of the 13C NMR chemical shifts. The CMAE and CRMSE values were in the ranges of 1.44 to 2.62 ppm and 1.72 to 3.53 ppm, respectively. The coefficients of determination (r2) were above 0.9976 for all tested functionals. C3 and C16 were consistently observed with the noticeable deviations ranged from 2.18 to 6.28 ppm and 2.39 to 4.98 ppm, respectively (Figure 2). The Nguyen Thi Nhu Y, Nguyen Trong Thien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 58-64 61 two best performers with strong correlations and low errors for 13C calculations were CAM- B3LYP (CMAE = 1.44 ppm, CRMSE = 1.72 ppm, and r2 = 0.9991), ωB97XD (CMAE = 1.48ppm, CRMSE = 1.80 ppm, and r2 = 0.9990). Table 1. 13C NMR chemical shifts of bakuchiol calculated using 11 functionals δ(13C) Entry Functional r2 CMAE CRMSE 1 B3LYP 0.9987 1.79 2.33 2 B3PW91 0.9988 1.97 2.49 3 BPV86 0.9978 2.36 3.17 4 CAM-B3LYP 0.9991 1.44 1.72 5 HCTH 0.9981 2.23 2.96 6 HSEH1PBE 0.9989 1.91 2.34 7 LSDA 0.9976 2.62 3.53 8 mPW1PW91 0.9989 1.91 2.36 9 PBEPBE 0.9989 1.91 2.34 10 TPSSTPSS 0.9981 2.50 2.94 11 ωB97XD 0.9990 1.48 1.80 Figure 2. Absolute deviations of 13C chemical shift calculations using 11 functionals. 3.2. The evaluation of 11 basis sets 11 Basic sets were employed for computing 13C chemical shifts of bakuchiol. In general, the calculated results were observed with low associated errors and strong linear correlations (r2 ≥ 0.9958). CMAE and CRMSE values were ranged from 1.79 to 4.97 ppm and 2.22 to 5.13 ppm, respectively (Table 3). The largest deviations were found for C3, C11, and C16 with CMAE and CRMSE values in the ranges of 1.05 to 6.25 ppm, 0.46 to 6.11 ppm, and 2.13 to 4.47 ppm, respectively (Figure 1). Nguyen Thi Nhu Y, Nguyen Trong Thien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 58-64 62 Table 2. The calculated 13C NMR chemical shifts of Bakuchiol in CHCl3 using 10 basic sets. All chemical shifts, CMAEs, and CRMSEs are in ppm. δ( 13C) Entry Basis set r 2 CMAE CRMSE 1 3-21G 0.9981 2.37 2.67 2 6-31G(d,p) 0.9987 1.79 2.33 3 6-31G(3d,p) 0.9971 2.62 3.21 4 6-31G(d,3p) 0.9975 2.33 2.67 5 6-31++G(d,p) 0.9958 3.35 3.43 6 6-311G 0.9976 1.93 2.71 7 DGDZVP 0.9985 2.19 2.22 8 DGDZVP2 0.9962 4.97 5.13 10 LANL2DZ 0.9970 3.13 3.28 11 LANL2MB 0.9970 3.80 3.81 Figure 3. Absolute deviations of 13C chemical shift calculations using 10 basis sets. 4. Conclusion We have performed the evaluation of 11 DFT functionals and 11 basis sets using GIAO method on the calculation of 13C chemical shifts for bakuchiol. Our results showed the two best performing functionals were CAM-B3LYP (CMAEs ≤ 1.44 ppm) and ωB97XD (CRMSEs ≤ 1.80 ppm), and the best basis set was 6- 31G(d,p) (CMAEs ≤ 1.79 ppm). In these cases, excellent correlations between theoretical and experimental data (r2 > 0.9987) were observed. Given such high degree of accuracy achieved in calculating 13C chemical shifts of bakuchiol, this work can be useful for supporting the assignments of the experimental NMR spectra of bakuchiol and similar retinoid compounds. Further studies on the chemical shift calculations of these compounds are under-investigation. Nguyen Thi Nhu Y, Nguyen Trong Thien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 58-64 63 References [1] S. Dhaliwal, I. Rybak, S.R. Ellis, M. Notay, M. Trivedi, W. Burney, A.R. Vaughn, M. Nguyen, P. Reiter, S. Bosanac, H. Yan, N. Foolad, R.K. Sivamani, Prospective, randomized, double‐blind assessment of topical bakuchiol and retinol for facial photoageing, Br. J. Dermatol. 180 (2019) 289–296. https://doi.org/10.1111/bjd.16918. [2] T. Esumi, C. Yamamoto, Y. Fukuyama, A short synthesis of (+)-bakuchiol, Synlett. 24 (2013) 1845– 1847. https://doi.org/10.1055/s-0033-1338968. [3] Z. Xin, X. Wu, T. Ji, B. Xu, Y. Han, M. Sun, S. Jiang, T. Li, W. Hu, C. Deng, Y. Yang, Bakuchiol: A newly discovered warrior against organ damage, Pharmacol. Res. 141 (2019) 208–213. https://doi.org/10.1016/j.phrs.2019.01.001. [4] H.D. Watts, M.N.A. Mohamed, J.D. Kubicki, Comparison of multistandard and TMS-standard calculated NMR shifts for coniferyl alcohol and application of the multistandard method to lignin dimers, J. Phys. Chem. B. 115 (2011) 1958–1970. https://doi.org/10.1021/jp110330q. [5] J.S. Lomas, 1 H NMR spectra of alcohols and diols in chloroform: DFT/GIAO calculation of chemical shifts, Magn. Reson. Chem. 52 (2014) 745–754. https://doi.org/10.1002/mrc.4130. [6] B.G. Diehl, H.D. Watts, J.D. Kubicki, M.R. Regner, J. Ralph, N.R. Brown, Towards lignin-protein crosslinking: Amino acid adducts of a lignin model quinone methide, Cellulose. 21 (2014) 1395–1407. https://doi.org/10.1007/s10570-014-0181-y. [7] K. Wolinski, J.F. Hinton, P. Pulay, Efficient Implementation of the Gauge-Independent Atomic Orbital Method for NMR Chemical Shift Calculations, J. Am. Chem. Soc. 112 (1990) 8251– 8260. https://doi.org/10.1021/ja00179a005. [8] J. Gauss, Effects of electron correlation in the calculation of nuclear magnetic resonance chemical shifts, J. Chem. Phys. 99 (1993) 3629–3643. https://doi.org/10.1063/1.466161. [9] R. Ditchfield, Self-consistent perturbation theory of diamagnetism I. A gauge-invariant LCAO method for N.M.R. Chemical shifts, Mol. Phys. 27 (1974) 789–807. https://doi.org/10.1080/00268977400100711. [10] M.A. Iron, Evaluation of the Factors Impacting the Accuracy of 13C NMR Chemical Shift Predictions using Density Functional Theory - The Advantage of Long-Range Corrected Functionals, J. Chem. Theory Comput. 13 (2017) 5798–5819. https://doi.org/10.1021/acs.jctc.7b00772. [11] M.J. Frisch, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, O. Farkas, J.B. Foresman, J. V. Ortiz, J. Cioslowski, F.D. J., Gaussian 09, Revision D.01, (2013). [12] J. Tomasi, B. Mennucci, R. Cammi, Quantum mechanical continuum solvation models, Chem. Rev. 105 (2005) 2999–3093. https://doi.org/10.1021/cr9904009. [13] J. Tomasi, B. Mennucci, E. Cancès, The IEF version of the PCM solvation method: An overview of a new method addressed to study molecular solutes at the QM ab initio level, in: J. Mol. Struct. THEOCHEM, Elsevier, 1999: pp. 211–226. https://doi.org/10.1016/S0166-1280(98)00553-3. [14] M.M. Francl, W.J. Pietro, W.J. Hehre, J.S. Binkley, M.S. Gordon, D.J. DeFrees, J.A. Pople, Self- consistent molecular orbital methods. XXIII. A polarization-type basis set for second-row elements, J. Chem. Phys. 77 (1982) 3654–3665. https://doi.org/10.1063/1.444267. [15] P.J. Stephens, F.J. Devlin, C.F. Chabalowski, M.J. Frisch, Ab Initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields, J. Phys. Chem. 98 (1994) 11623–11627. https://doi.org/10.1021/j100096a001. [16] A.D. Becke, Density-functional thermochemistry. III. The role of exact exchange, J. Chem. Phys. 98 (1993) 5648–5652. https://doi.org/10.1063/1.464913. [17] A.D. Becke, Density-functional exchange-energy approximation with correct asymptotic behavior, Phys. Rev. A. 38 (1988) 3098–3100. https://doi.org/10.1103/PhysRevA.38.3098. [18] J.P. Perdew, J.A. Chevary, S.H. Vosko, K.A. Jackson, M.R. Pederson, D.J. Singh, C. Fiolhais, Atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation, Phys. Rev. B. 46 (1992) 6671–6687. https://doi.org/10.1103/PhysRevB.46.6671. [19] J.P. Perdew, J.A. Chevary, S.H. Vosko, K.A. Jackson, M.R. Pederson, D.J. Singh, C. Fiolhais, Erratum: Atoms, molecules, solids, and surfaces: Nguyen Thi Nhu Y, Nguyen Trong Thien / Tạp chí Khoa học và Công nghệ Đại học Duy Tân 02(45) (2021) 58-64 64 Applications of the generalized gradient approximation for exchange and correlation (Physical Review B (1993) 48, 7, (4978)), Phys. Rev. B. 48 (1993) 4978. https://doi.org/10.1103/PhysRevB.48.4978.2. [20] J.P. Perdew, Density-functional approximation for the correlation energy of the inhomogeneous electron gas, Phys. Rev. B. 33 (1986) 8822–8824. https://doi.org/10.1103/PhysRevB.33.8822. [21] S.H. Vosko, L. Wilk, M. Nusair, Accurate spin- dependent electron liquid correlation energies for local spin density calculations: a critical analysis, Can. J. Phys. 58 (1980) 1200–1211. https://doi.org/10.1139/p80-159. [22] T. Yanai, D.P. Tew, N.C. Handy, A new hybrid exchange-correlation functional using the Coulomb- attenuating method (CAM-B3LYP), Chem. Phys. Lett. 393 (2004) 51–57. https://doi.org/10.1016/j.cplett.2004.06.011. [23] F.A. Hamprecht, A.J. Cohen, D.J. Tozer, N.C. Handy, Development and assessment of new exchange-correlation functionals, J. Chem. Phys. 109 (1998) 6264–6271. https://doi.org/10.1063/1.477267. [24] A. Daniel Boese, N.L. Doltsinis, N.C. Handy, M. Sprik, New generalized gradient approximation functionals, J. Chem. Phys. 112 (2000) 1670–1678. https://doi.org/10.1063/1.480732. [25] A.D. Boese, N.C. Handy, A new parametrization of exchange-correlation generalized gradient approximation functionals, J. Chem. Phys. 114 (2001) 5497–5503. https://doi.org/10.1063/1.1347371. [26] J. Heyd, G.E. Scuseria, M. Ernzerhof, Hybrid functionals based on a screened Coulomb potential, J. Chem. Phys. 118 (2003) 8207–8215. https://doi.org/10.1063/1.1564060. [27] M. Ernzerhof, J.P. Perdew, Generalized gradient approximation to the angle- and system-averaged exchange hole, J. Chem. Phys. 109 (1998) 3313– 3320. https://doi.org/10.1063/1.476928. [28] J.P. Perdew, Y. Wang, Accurate and simple analytic representation of the electron-gas correlation energy, Phys. Rev. B. 45 (1992) 13244–13249. https://doi.org/10.1103/PhysRevB.45.13244. [29] J.P. Perdew, K. Burke, Generalized gradient approximation for the exchange-correlation hole of a many-electron system, Phys. Rev. B - Condens. Matter Mater. Phys. 54 (1996) 16533–16539. https://doi.org/10.1103/PhysRevB.54.16533. [30] C. Adamo, V. Barone, Exchange functionals with improved long-range behavior and adiabatic connection methods without adjustable parameters: The mPW and mPW1PW models, J. Chem. Phys. 108 (1998) 664–675. https://doi.org/10.1063/1.475428. [31] J.P. Perdew, K. Burke, M. Ernzerhof, Generalized gradient approximation made simple, Phys. Rev. Lett. 77 (1996) 3865–3868. https://doi.org/10.1103/PhysRevLett.77.3865. [32] J. Tao, J.P. Perdew, V.N. Staroverov, G.E. Scuseria, Climbing the density functional ladder: Nonempirical meta–generalized gradient approximation designed for molecules and solids, Phys. Rev. Lett. 91 (2003) 146401. https://doi.org/10.1103/PhysRevLett.91.146401. [33] J.P. Perdew, A. Ruzsinszky, G.I. Csonka, L.A. Constantin, J. Sun, Workhorse semilocal density functional for condensed matter physics and quantum chemistry, Phys. Rev. Lett. 103 (2009) 026403. https://doi.org/10.1103/PhysRevLett.103.026403. [34] J. Da Chai, M. Head-Gordon, Systematic optimization of long-range corrected hybrid density functionals, J. Chem. Phys. 128 (2008) 084106. https://doi.org/10.1063/1.2834918. [35] J. Chai, M. Head-Gordon, Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections, Phys. Chem. Chem. Phys. 10 (2008) 6615–6620. https://doi.org/10.1039/B810189B. [36] J. Stephen Binkley, J. A. Pople, W. J. Hehre, Self- consistent molecular orbital methods. 21. Small split-valence basis sets for first-row elements, J. Am. Chem. Soc. 102 (1980) 939–947. https://doi.org/10.1021/ja00523a008. [37] R. Ditchfield, W.J. Hehre, J.A. Pople, Self- consistent molecular-orbital methods. IX. An extended gaussian-type basis for molecular-orbital studies of organic molecules, J. Chem. Phys. 54 (1971) 720–723. https://doi.org/10.1063/1.1674902. [38] C. Sosa, J. Andzelm, B. C. Elkin, E. Wimmer, K. D. Dobbs, D. A. Dixon, A local density functional study of the structure and vibrational frequencies of molecular transition-metal compounds, J. Phys. Chem. 96 (1992) 6630–6636. https://doi.org/10.1021/j100195a022. [39] P.J. Hay, W.R. Wadt, Ab initio effective core potentials for molecular calculations. Potentials for the transition metal atoms Sc to Hg, J. Chem. Phys. 82 (1985) 270–283. https://doi.org/10.1063/1.448799. [40] W.R. Wadt, P.J. Hay, Ab initio effective core potentials for molecular calculations. Potentials for main group elements Na to Bi, J. Chem. Phys. 82 (1985) 284–298. https://doi.org/10.1063/1.448800. [41] T.T. Nguyen, P.Q. Le, J. Helminen, J. Sipilä, The 1H and 13C chemical shifts of 5–5 lignin model dimers: An evaluation of DFT functionals, J. Mol. Struct. 1226 (2021) 129300. https://doi.org/10.1016/j.molstruc.2020.129300.
File đính kèm:
 tinh_toan_do_dich_chuyen_hoa_hoc_13c_cua_bakuchioltac_nhan_m.pdf
tinh_toan_do_dich_chuyen_hoa_hoc_13c_cua_bakuchioltac_nhan_m.pdf

