Alkaloids isolated from Hippeastrum reticulatum (L’Hér.) Herb. and their acetylcholinesterase inhibitory activities
Background: Hippeastrum reticulatum (L.Hér.) Herb is a species of the Hippeastrum Herb. genus.
Screening studies have shown that this species has the ability to inhibit the enzyme acetylcholinesterase.
So far, research on this species is still very limited. The purpose of this study is to provide some more
informations about the chemical composition and bioactive of isolated compounds from this species.
Materials and method: Bulbs of Hippeastrum reticulatum was collected in Thua Thien Hue province in May
2018. The compounds were isolated by using various chromatographic methods and their structures were
identified by 1D and 2D-NMR spectroscopic methods in reference to the literature. The acetylcholinesterase
inhibitory activity was determined by Ellman’s microplate colorimetric method. Results and conclusions:
Two alkaloids including N-methyltiramine (1), narciclasine-4-O-β-D-xylopyranoside (2) was isolated from
bulbs of Hippeastrum reticulatum (L’Hér.) Herb. These compounds were isolated from Hippeastrum Herb.
genus for the first time. Compound 2 showed moderate acetylcholinesterase inhibitory activity, with IC50
value of 70.06 ± 1.46 µg/mL

Trang 1
Trang 2
Trang 3

Trang 4

Trang 5
Tóm tắt nội dung tài liệu: Alkaloids isolated from Hippeastrum reticulatum (L’Hér.) Herb. and their acetylcholinesterase inhibitory activities

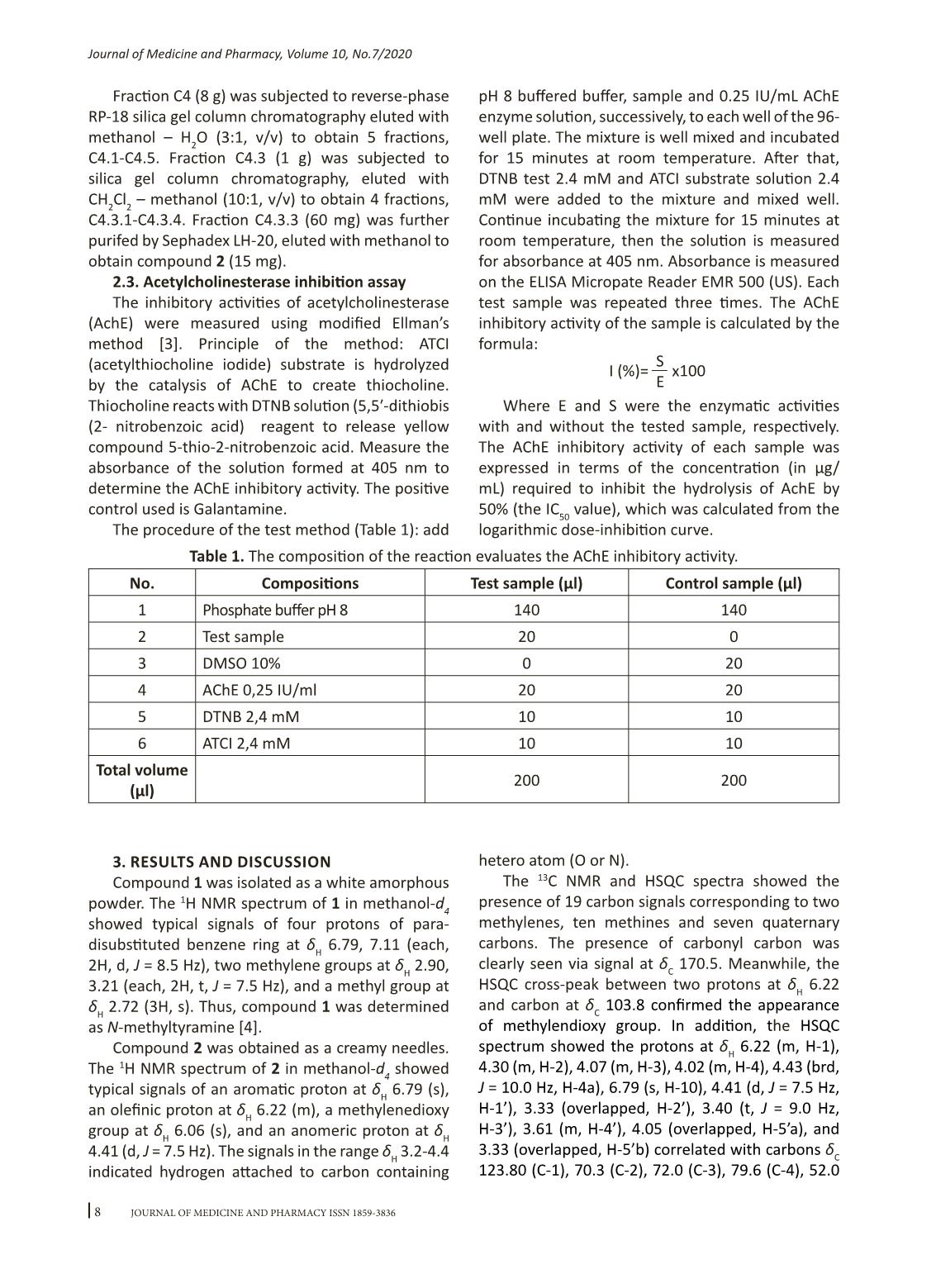
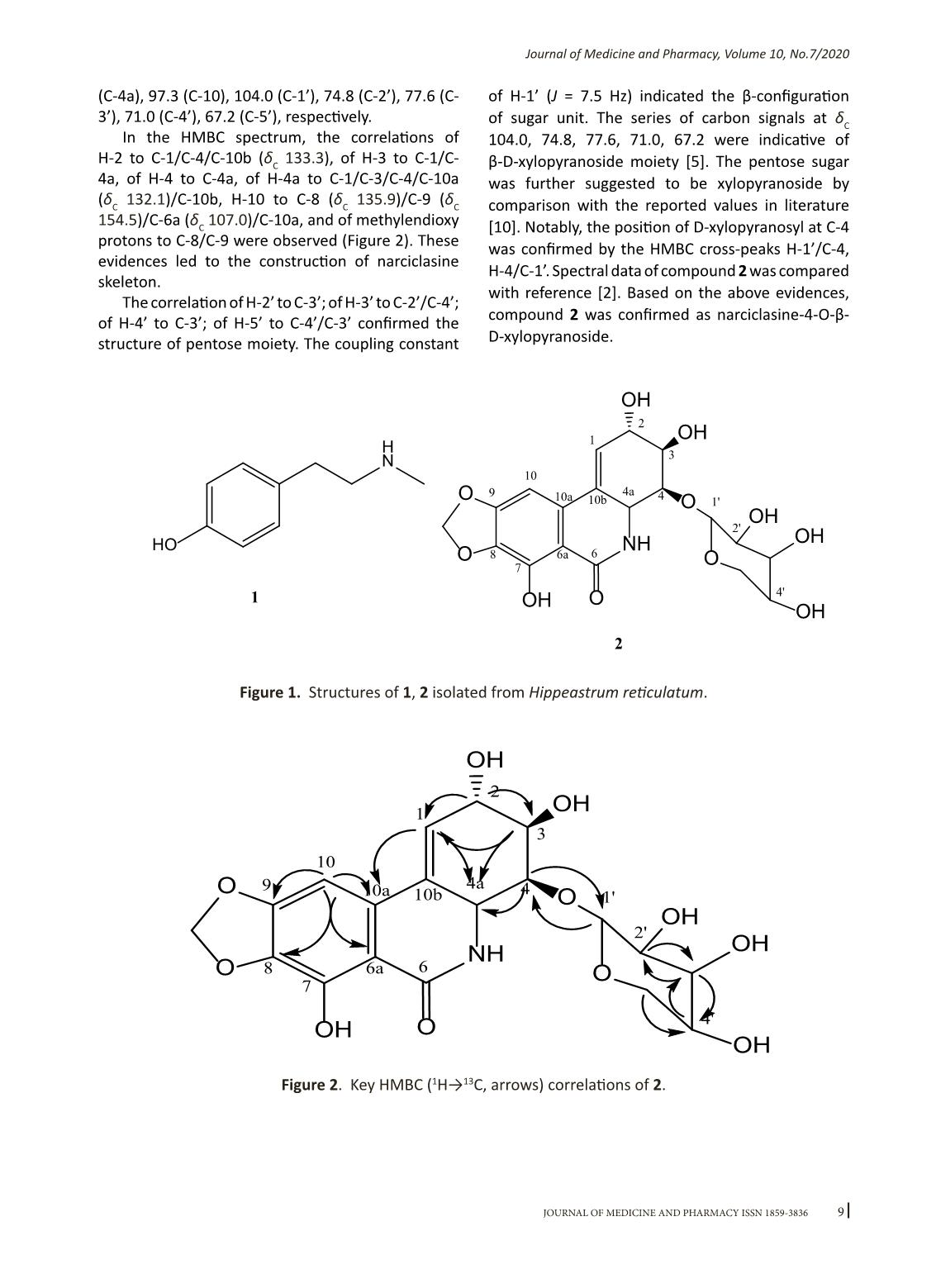
7Journal of Medicine and Pharmacy, Volume 10, No.7/2020 Alkaloids isolated from Hippeastrum reticulatum (L’Hér.) Herb. and their acetylcholinesterase inhibitory activities Hoang Xuan Huyen Trang, Ho Viet Duc, Nguyen Thi Hoai Faculty of Pharmacy, Hue University of Medicine and Pharmacy, Hue University Abstract Background: Hippeastrum reticulatum (L.Hér.) Herb is a species of the Hippeastrum Herb. genus. Screening studies have shown that this species has the ability to inhibit the enzyme acetylcholinesterase. So far, research on this species is still very limited. The purpose of this study is to provide some more informations about the chemical composition and bioactive of isolated compounds from this species. Materials and method: Bulbs of Hippeastrum reticulatum was collected in Thua Thien Hue province in May 2018. The compounds were isolated by using various chromatographic methods and their structures were identified by 1D and 2D-NMR spectroscopic methods in reference to the literature. The acetylcholinesterase inhibitory activity was determined by Ellman’s microplate colorimetric method. Results and conclusions: Two alkaloids including N-methyltiramine (1), narciclasine-4-O-β-D-xylopyranoside (2) was isolated from bulbs of Hippeastrum reticulatum (L’Hér.) Herb. These compounds were isolated from Hippeastrum Herb. genus for the first time. Compound 2 showed moderate acetylcholinesterase inhibitory activity, with IC50 value of 70.06 ± 1.46 µg/mL. Keywords: Hippeastrum reticulatum (L’Hér.) Herb., alkaloid, N-methyltiramine, narciclasine-4-O-β-D- xylopyranoside. Corresponding author: Nguyen Thi Hoai, email: nthoai@huemed-univ.edu.vn DOI: 10.34071/jmp.2020.7.1 Received: 17/12/2019, Accepted: 23/3/2020 1. INTRODUCTION Hippeastrum Herb. is a large genus of the Amaryllidaceae family of more than 90 species recorded. The species belong to this genus possess several important biological activities, such as antibacterial, antioxidant, antiviral, acetylcholinesterase inhibitors... In Vietnam, this genus has 2 species: Hippeastrum equestre and Hippeastrum reticulatum. Screening studies indicated that both species have a potent inhibitory effects of acetylcholinesterase, particularly Hippeastrum reticulatum. However, there are still few studies on Hippeastrum reticulatum species in Vietnam so far. The aim of this study is to contribute knowledge to the chemical composition and bioactivity of isolated compounds from Hippeastrum reticulatum. 2. MATERIALS AND METHODS 2.1. Plant materials The bulb of Hippeastrum reticulatum (L’Hér.) Herb. was picked up in May 2018 in Thua Thien Hue province, Vietnam. Its identify was confirmed by Dr. Vu Tien Chinh, Vietnam National Museum of Nature, the Vietnam Academy of Science and Technology. 2.2. Extraction and isolation The bulb of Hippeastrum reticulatum (L’Hér.) Herb. was washed, dried at 50oC (12.5 kg) then powdered into powder, extracted with methanol (20 L × 3 times) by immersion at room temperature to yield extract. This extract was subjected to Diaion HP-20 column chromatography. Pass water through the column to remove water-soluble components, then elute the compounds with methanol to obtain a methanol extract (150 g). The methanol extract was acidified with 2% HCl to pH 2 and then extracted with ethyl acetate (1 L × 3 times) to obtain ethyl acetate (EtOAc) fraction (60 g). The remaining acid solution was alkalined with NH3 to pH 10 and then extracted with dichloromethan (CH2Cl2) (1L × 3 times) to obtain C fraction (30 g). The C fraction (30g) was subjected to silica gel column chromatography, eluted with CH2Cl2 – methanol – H2O (5:1:0.1, v/v/v) to obtain 5 fractions, C1-C5. Fraction C1 (4g) was subjected to reverse- phase RP-18 silica gel column chromatography eluted with aceton – H2O (5:1, v/v) to obtain 6 fractions, C1.1-C1.6. Fraction C1.3 (600mg) was subjected to silica gel chromatography, eluted with CH2Cl2-methanol-NH3 (10:1:0.1, v/v/v) to obtain 6 fractions, C1.3.1-C1.3.6. Fraction C1.3.2 (120mg) was subjected to reverse-phase RP-18 silica gel column chromatography, eluted with methanol-H2O (3:1, v/v) to obtain compound 1 (8 mg). 8Journal of Medicine and Pharmacy, Volume 10, No.7/2020 Fraction C4 (8 g) was subjected to reverse-phase RP-18 silica gel column chromatography eluted with methanol – H2O (3:1, v/v) to obtain 5 fractions, C4.1-C4.5. Fraction C4.3 (1 g) was subjected to silica gel column chromatography, eluted with CH2Cl2 – methanol (10:1, v/v) to obtain 4 fractions, C4.3.1-C4.3.4. Fraction C4.3.3 (60 mg) was further purifed by Sephadex LH-20, eluted with methanol to obtain compound 2 (15 mg). 2.3. Acetylcholinesterase inhibition assay The inhibitory activities of acetylcholinesterase (AchE) were measured using modified Ellman’s method [3]. Principle of the method: ATCI (acetylthiocholine iodide) substrate is hydrolyzed by the catalysis of AChE to create thiocholine. Thiocholine reacts with DTNB solution (5,5′-dithiobis (2- nitrobenzoic acid) reagent to release yellow compound 5-thio-2-nitrobenzoic acid. Measure the absorbance of the solution formed at 405 nm to determine the AChE inhibitory activity. The positive control used is Galantamine. The procedure of the test method (Table 1): add pH 8 buffered buffer, sample and 0.25 IU/mL AChE enzyme solution, successively, to each well of the 96- well plate. The mixture is well mixed and incubated for 15 minutes at room temperature. After that, DTNB test 2.4 mM and ATCI substrate solution 2.4 mM were added to the mixture and mixed well. Continue incubating the mixture for 15 minutes at room temperature, then the solution is measured for absorbance at 405 nm. Absorbance is measured on the ELISA Micropate Reader EMR 500 (US). Each test sample was repeated three times. The AChE inhibitory activity of the sample is calculated by the formula: Where E and S were the enzymatic activities with and without the tested sample, respectively. The AChE inhibitory activity of each sample was expressed in terms of the concentration (in µg/ mL) required to inhibit the hydrolysis of AchE by 50% (the IC50 value), which was calculated from the logarithmic dose-inhibition curve. I (%)= x100 S E Table 1. The composition of the reaction evaluates the AChE inhibitory activity. No. Compositions Test sample (µl) Control sample (µl) 1 Phosphate buffer pH 8 140 140 2 Test sample 20 0 3 DMSO 10% 0 20 4 AChE 0,25 IU/ml 20 20 5 DTNB 2,4 mM 10 10 6 ATCI 2,4 mM 10 10 Total volume (µl) 200 200 3. RESULTS AND DISCUSSION Compound 1 was isolated as a white amorphous powder. The 1H NMR spectrum of 1 in methanol-d 4 showed typical signals of four protons of para- disubstituted benzene ring at δH 6.79, 7.11 (each, 2H, d, J = 8.5 Hz), two methylene groups at δH 2.90, 3.21 (each, 2H, t, J = 7.5 Hz), and a methyl group at δH 2.72 (3H, s). Thus, compound 1 was determined as N-methyltyramine [4]. Compound 2 was obtained as a creamy needles. The 1H NMR spectrum of 2 in methanol-d 4 showed typical signals of an aromatic proton at δH 6.79 (s), an olefinic proton at δH 6.22 (m), a methylenedioxy group at δH 6.06 (s), and an anomeric proton at δH 4.41 (d, J = 7.5 Hz). The signals in the range δH 3.2-4.4 indicated hydrogen attached to carbon containing hetero atom (O or N). The 13C NMR and HSQC spectra showed the presence of 19 carbon signals corresponding to two methylenes, ten methines and seven quaternary carbons. The presence of carbonyl carbon was clearly seen via signal at δC 170.5. Meanwhile, the HSQC cross-peak between two protons at δH 6.22 and carbon at δC 103.8 confirmed the appearance of methylendioxy group. In addition, the HSQC spectrum showed the protons at δH 6.22 (m, H-1), 4.30 (m, H-2), 4.07 (m, H-3), 4.02 (m, H-4), 4.43 (brd, J = 10.0 Hz, H-4a), 6.79 (s, H-10), 4.41 (d, J = 7.5 Hz, H-1’), 3.33 (overlapped, H-2’), 3.40 (t, J = 9.0 Hz, H-3’), 3.61 (m, H-4’), 4.05 (overlapped, H-5’a), and 3.33 (overlapped, H-5’b) correlated with carbons δC 123.80 (C-1), 70.3 (C-2), 72.0 (C-3), 79.6 (C-4), 52.0 9Journal of Medicine and Pharmacy, Volume 10, No.7/2020 (C-4a), 97.3 (C-10), 104.0 (C-1’), 74.8 (C-2’), 77.6 (C- 3’), 71.0 (C-4’), 67.2 (C-5’), respectively. In the HMBC spectrum, the correlations of H-2 to C-1/C-4/C-10b (δC 133.3), of H-3 to C-1/C- 4a, of H-4 to C-4a, of H-4a to C-1/C-3/C-4/C-10a (δC 132.1)/C-10b, H-10 to C-8 (δC 135.9)/C-9 (δC 154.5)/C-6a (δC 107.0)/C-10a, and of methylendioxy protons to C-8/C-9 were observed (Figure 2). These evidences led to the construction of narciclasine skeleton. The correlation of H-2’ to C-3’; of H-3’ to C-2’/C-4’; of H-4’ to C-3’; of H-5’ to C-4’/C-3’ confirmed the structure of pentose moiety. The coupling constant of H-1’ (J = 7.5 Hz) indicated the β-configuration of sugar unit. The series of carbon signals at δC 104.0, 74.8, 77.6, 71.0, 67.2 were indicative of β-D-xylopyranoside moiety [5]. The pentose sugar was further suggested to be xylopyranoside by comparison with the reported values in literature [10]. Notably, the position of D-xylopyranosyl at C-4 was confirmed by the HMBC cross-peaks H-1’/C-4, H-4/C-1’. Spectral data of compound 2 was compared with reference [2]. Based on the above evidences, compound 2 was confirmed as narciclasine-4-O-β- D-xylopyranoside. Figure 1. Structures of 1, 2 isolated from Hippeastrum reticulatum. Figure 2. Key HMBC (1H→13C, arrows) correlations of 2. O O NH OH OH O O OH OH OH OOH 1 2 3 44a 1' 2' 4' 66a 7 9 10 10a 10b 8 H N HO 2 O O NH OH OH O O OH OH OH OOH 1 2 3 44a 1' 2' 4' 66a 7 9 10 10a 10b 8 1 10 Journal of Medicine and Pharmacy, Volume 10, No.7/2020 Table 2. 1H (500 MHz) and 13C (125 MHz) NMR data of 2 in methanol-d4 [δ (ppm), J (Hz)] Position δC δH 1 123.8 6.22 t (4.0, 6.5) 2 70.3 4.30 m 3 72.0 4.07 m 4 79.6 4.02 m 4a 52.1 4.43 m 6 170.5 - 6a 107.0 - 7 146.0 - 8 135.9 - 9 154.5 - 10a 132.1 - 10b 133.3 - 10 97.3 6.79 s 1’ 104.0 4.41 (d, 7.5) 2’ 74.8 3.33 m 3’ 77.7 3.39 m 4’ 71.1 3.61 m 5’ 67.3 4.05 m -OCH2O- 103.8 6.06 brs The AChE inhibitory activities of the isolated compounds were assessed at various concentrations. Galanthamine was used as a positive control. The results are summarized in Table 3. Results showed that compound 2 did not show the ability to inhibit AChE at the test concentration (500 µg / mL). Compound 1 have a moderate AChE inhibiting effect with IC50 values of 70,06 ± 1,46. N-Methyltyramine (1) has been isolated from a variety of plant species such as Coryphantha missouriensis (Cactaceae) [6], Pilosocereus maxonii (Rose) [7], and Acacia schweinfurthii (Leguminosae) [9]. Previous studies have shown that N-methyltyramine (1) increases blood pressure in the anaesthetized rat, relaxes guinea pig ileum and increases both the force and rate of contraction of guinea-pig right atrium by inducing the release of noradrenaline [1]. However, this is the first report of AChE inhibition activity of N-methyltyramine (1). Table 3. In vitro AChE inhibitory activities of compounds 1 and 2 Compounds IC50 (µg/mL) N-methyltyramine (1) 70.06 ± 1.46 Narciclasine-4-O-β-D-xylopyranoside (2) > 500 Galantamine 0.33 ± 0.01 4. CONCLUSION Two alkaloids, N-methyltiramine (1), narciclasine- 4-O-β-D-xylopyranoside (2), were isolated from the bulb of Hippeastrum reticulatum and identified by comparison of their 1D and 2D NMR spectroscopic with those reported in the literature. These compounds were isolated from Hippeastrum Herb. genus for the first time. Compound 1 showed moderate inhibitory activities against AChE, with IC50 values of 70.06 ± 1.46 µg/mL. 11 Journal of Medicine and Pharmacy, Volume 10, No.7/2020 1. Christine S.Evans, E.Arthur Bell, E.Stewart Johnson (1979), “N-methyltyramine, a biologically active amine in Acacia seeds”, Phytochemistry, 18 (12), 2022–2023. 2. Deepali Katoch, Dharmesh Kumar, Yogendra S Padwad, Bikram Singh & Upendra Sharma (2019), “Narciclasine-4-O-β-D-xylopyranoside, a new narciclasine glycoside from Zephyranthes minuta”, Natural Product Research, 1–8. 3. G.L. Ellman, K.D. Courtney, V. Andres Jr., R.M. Featherstone (1961), “A new and rapid colorimetric determination of acetylcholinesterase activity”, Biochemical Pharmacology, 7, 88–95. 4. Hiroyuki Kikuchi, Nahoko Uchiyama, Jun Ogata, Ruri Kikura-Hanajiri, Yukihiro Goda (2010), “Chemical constituents and DNA sequence analysis of a psychotropic herbal product”, Forensic Toxicol, 28, 77–83. 5. Pan ZH, Li Y, Liu JL, Ning DS, Li DP,Wu XD, Wen YX. (2012), “A cytotoxic cardenolide and a saponin from the rhizomes of Tupistra chinensis”, Fitoterapia, 83, 1489– 1493. 6. Pummangura S, McLaughlin JL (1981), “Cactus alkaloids. XLVII. β-Phenethylamines from the “Missouri Pincushion”, Coryphantha (Neobessya) missouriensis”, Journal of Natural Products, 44, 614–616. 7. Pummangura S, Nichols DE, McLaughlin JL (1977), “Cactus alkaloids XXXIII: β-phenethylamines from the Guatemalan cactus Pilosocereus maxonii”, Journal of Pharmaceutical Sciences, 66, 1485–1487 8. Surup F, Wiebach V, Kuhnert E, Stadler M. (2016), “Truncaquinones A and B, asterriquinones from Annulohypoxylon truncatum”, Tetrahedron Letter, 57, 2183–2185. 9. Vanderveen RL, West LG, McLaughlin JL (1974), “N-Methyltyramine from Opuntia clavata”, Phytochemistry, 13, 866–867. 10. Zhang ZQ, Chen JC, Zhang XM, Li ZR, Qui MH (2008), “Two new spirostanol saponins from Reineckia carnea”, Helvetica Chimica Acta, 91, 1494–1509. REFERENCES
File đính kèm:
 alkaloids_isolated_from_hippeastrum_reticulatum_lher_herb_an.pdf
alkaloids_isolated_from_hippeastrum_reticulatum_lher_herb_an.pdf

