A phytochemical study of the ethyl acetate extract (EtOAc) of Styrax annamensis leaves resulted in the isolation and determination of six known compounds, including three nor-Neolignans type 2-phenylbenzofurans egonol (1), egonoic acid (2) and (–)-machice
Spinasterol (1) and four flavonoids (2-5) were isolated from Albizia myriophylla wood collected at Phu Yen
province, Vietnam. The chemical structures of these compounds were elucidated by NMR spectroscopy as well as
compared with data in the literature. The contents of some high yield isolated compounds (2-4) in the ethanol extract of
A. myriophylla were quantified by high-performance liquid chromatography. These HPLC chromatograms could be
used to reveal the presence of isolated compounds as markers of the sample.

Trang 1

Trang 2
Trang 3

Trang 4

Trang 5
Bạn đang xem tài liệu "A phytochemical study of the ethyl acetate extract (EtOAc) of Styrax annamensis leaves resulted in the isolation and determination of six known compounds, including three nor-Neolignans type 2-phenylbenzofurans egonol (1), egonoic acid (2) and (–)-machice", để tải tài liệu gốc về máy hãy click vào nút Download ở trên
Tóm tắt nội dung tài liệu: A phytochemical study of the ethyl acetate extract (EtOAc) of Styrax annamensis leaves resulted in the isolation and determination of six known compounds, including three nor-Neolignans type 2-phenylbenzofurans egonol (1), egonoic acid (2) and (–)-machice

Cite this paper: Vietnam J. Chem., 2020, 58(5), 597-601 Article
DOI: 10.1002/vjch.202000018
597 Wiley Online Library © 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH
Chemical constituents of Albizia myriophylla wood and the HPLC
determination of some high yield compounds as markers
Pham Thi Tam1, Phung Van Trung2, Vo Thi Nga3, Nguyen Thi Anh Tuyet4,
Nguyen Kim Phi Phung5, Ngo Thi Thuy Duong5, Nguyen Thi Hoai Thu6*
1Sai Gon University, 273, An Duong Vuong, Dist. 5, Ho Chi Minh City 70000, Viet Nam
2Center for Research and Technology Transfer, Vietnam Academy of Science and Technology,
18, Hoang Quoc Viet, Cau Giay, Hanoi 10000, Viet Nam
3University of Technology and Education Ho Chi Minh City, 1, Vo Van Ngan, Thu Duc, Ho Chi Minh City
70000, Viet Nam
4University of Education Ho Chi Minh City, 280, An Duong Vuong, Dist. 5, Ho Chi Minh City 70000,
Viet Nam
5University of Science, Vietnam National University - Ho Chi Minh City, 227, Nguyen Van Cu,
Dist.5, Ho Chi Minh City 70000, Viet Nam
6University of Medicine and Pharmacy at Ho Chi Minh City, 217, Hong Bang, Dist. 5, Ho Chi Minh City
70000, Viet Nam
Submitted February 21, 2020; Accepted March 19, 2020
Abstract
Spinasterol (1) and four flavonoids (2-5) were isolated from Albizia myriophylla wood collected at Phu Yen
province, Vietnam. The chemical structures of these compounds were elucidated by NMR spectroscopy as well as
compared with data in the literature. The contents of some high yield isolated compounds (2-4) in the ethanol extract of
A. myriophylla were quantified by high-performance liquid chromatography. These HPLC chromatograms could be
used to reveal the presence of isolated compounds as markers of the sample.
Keywords. Albizia myriophylla, flavonoid, content determination, HPLC.
1. INTRODUCTION
Albizia myriophylla is widely distributed in Asia
such as India, Laos, Cambodia, Myanmar, Thailand
and Vietnam etc.[1] In Vietnamese traditional
medicine, it was used to cure cough, bronchitis,
toothache, obesity, dyspepsia, diabetes, cancer etc.[1-
3] There were many researches about chemical
constituents and bioactivities of Albizia
myriophylla.[2-5] However, in Vietnam, there has
been no chemical study on this plant although it is
now cultivated at Phu Yen province for the internal
commercial use thanks to its above-mentioned
medicinal value.
In this paper, we report the isolation of five
compounds, spinasterol (1), 7,3',4'-trihydroxy-3-
methoxyflavone (2), fisetin (3), (2S)-7,3',4'-
trihydroxyflavanone (4) and (2S,3S,4S)-7,3,4,3',4'-
pentahydroxyflavane (5). The contents of some high
yield compounds (2, 3, 4) were determined by HPLC.
2. MATERIALS AND METHODS
2.1. General experimental procedures
The 1H-NMR (500 MHz) and 13C-NMR (125 MHz)
spectra were recorded on a Bruker AM500 FT-NMR
spectrometer. LR-MS spectra were recorded by
UPLC-MS (Ultimate RS 3000 MSQ Pluc. Thermo,
USA. Optical rotations were measured on a Kruss
(Germany) polarimeter with the length of the tube of
2 decimetres. HPLC analyses were carried out on a
HPLC Hewlett Packard 1050 series system include a
Quartenary pump, an Autosampler, a Thermostatted
column and a DAD detector. All moduls and the
data processing were controlled by Chemstation
software (Ver. A.10). The column was Phenomenex
Vietnam Journal of Chemistry Nguyen Thi Hoai Thu et al.
© 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 598
LUNA 5u C18 (250×4.6 mm, 5 m). The mobile
phase was composed of methanol (A) and aqueous
formic acid 0.1 % (B): from 0-18 min, 35 % A, 65 %
B; from 19-23 min, 70 % A, 30 % B; from 24-30
min, 100 % A, 0 % B; at a flow rate 1.0 mL/min.
Injection volume was of 10 L. All sample and pure
compounds were filtered by 0.45 m membrane
before injection.
2.2. Plant material
Albizia myriophylla wood sample was collected at
Hoa Hiep Nam industrial zone, Dong Hoa district,
Phu Yen province, Vietnam in June, 2018. This was
the donation of Mr. Hoang Xuan Lam, Middle
Vietnam Research and Manufacturing Organic
Medicinal Herb Centre in Phu Yen province.
2.3. Extraction and isolation
The dried powder wood (3.1 kg) of A. myriophylla
was exhaustedly extracted with ethanol by
maceration at room temperature. After evaporating,
the ethanolic filtrated solution gave crude extract
(210 g). This crude was chromatographed on silica
gel and successively eluted with n-hexane, ethyl
acetate and methanol to afford three fractions,
respectively. The ethyl acetate extract was
chromatographed, eluted with n-hexane:ethyl acetate
(20:80, 0:100), ethyl acetate:methanol (90:10, 80:20,
70:30, 0:100) to give 5 sub-fractions, EA1 EA5.
The sub-fraction EA2 (7.0 g) was applied to a silica
gel column, and eluted with n-hexane:ethyl acetate
(96:4) to afford 1 (15 mg). The same procedure for
the sub-fraction EA3 (5.0 g) was carried out, eluted
with n-hexane:ethyl acetate (60:40) to obtain 2 (110
mg), 3 (24 mg), 4 (15 mg) and 5 (8.5 mg).
Spinasterol (1): white powder. ESI-LRMS: m/z
473.6 [M+CH3COOH+H]+. 1H-NMR (CDCl3,
ppm, J in Hertz): 3.59 (1H, m, H3), 5.15 (1H, brs,
H7), 0.55 (3H, s, H18), 0.80 (3H, s, H19), 1.02 (3H,
d, 6.5, H21), 5.16 (1H, dd, 14.5, 9.0, H22), 5.03 (1H,
dd, 15.0, 8.5, H23), 0.85 (3H, d, 6.0, H26), 0.80 (3H,
d, 6.0, H27), 0.81 (3H, t, 7.0, H29). 13C-NMR
(CDCl3, ppm): 37.1 (C1), 31.4 (C2), 71.0 (C3),
37.9 (C4), 40.2 (C5), 29.5 (C6), 117.4 (C7), 139.5
(C8), 49.4 (C9), 34.1 (C10), 21.5 (C11), 39.4 (C12),
43.2 (C13), 55.0 (C14), 22.9 (C15), 28.4 (C16), 55.8
(C17), 11.9 (C18), 12.9 (C19), 40.7 (C20), 21.3
(C21), 138.1 (C22), 129.4 (C23), 51.2 (C24), 31.8
(C25), 21.0 (C26), 18.9 (C27), 25.3 (C28), and 12.1
(C29).
7,3',4'-Trihydroxy-3-methoxyflavone (2):
yellow crystal. ESI-LRMS m/z 301.8 [M+H]+. 1H-
NMR (CDCl3, ppm, J in He ... LRMS m/z 271.1 [M-H]-. [α]D
25 -22.7 (c
0.75, EtOAc). 1H-NMR (CDCl3, ppm, J in Hertz):
5.23 (1H, dd, 13.0, 2.0, H2), 2.93 (1H, dd, 16.5,
13.0, H3a), 2.66 (1H, dd, 16.5, 2.5, H3b), 7.70 (1H,
d, 8.5, H5), 6.44 (1H, dd, 8.5, 1.5, H6), 6.31 (1H, d,
1.5, H8), 6.87 (1H, d, 2.0, H2 ), 6.77 (1H, d, 8.5,
H5 ), 6.74 (1H, dd, 8.5, 2.0, H6 ). 13C-NMR (CDCl3)
data, see table 2.
Table 1: The contents of some isolated compounds
in the ethanol extract of A. myriophylla
Compound
Rt
(min)
Intergration
C
(ppm)
m
(mg)
a
(%)
Content
(mg/g)
2 14.299 299.312 11.62 121.9 15 1.12
3 14.105 49.500 4.354 121.9 15 0.42
4 12.083 581.433 12.60 121.9 15 1.22
C: Concentration of the compound;
m: Mass of the ethanol extract;
a: Humidity of the ethanol extract.
(2S,3S,4S)-7,3,4,3',4'-Pentahydroxyflavane
(5): brown powder. ESI-LRMS m/z 289.1 [M-H]-.
[α]D
25 +38.9 (c 4.25, MeOH). 1H-NMR (CD3OD, 500
MHz, ppm, J in Hertz): 5.02 (1H, brs, H2), 3.99
(1H, d, 4.0, H3), 4.95 (1H, d, 4.0, H4), 7.32 (1H, d,
8.5, H5), 6.45 (1H, dd, 8.5, 2.5, H6), 6.32 (1H, d,
2.0, H8), 7.05 (1H, d, 1.5, H2 ), 6.80 (1H, d, 8.5,
H5 ), 6.86 (1H, dd, 8.0, 1.5, H6 ). 13C-NMR
(CD3OD, 125 MHz) data, see table 2.
2.4. HPLC determination of major compounds in
the ethanol extract of A. myriophylla
Compounds 2, 3, 4 were studied because of their
good isolated amount and bioactivities.[5] They were
used as standards with the purity of about 98 %.
Vietnam Journal of Chemistry Chemical constituents of Albizia myriophylla
© 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 599
Establish of standard curve of pure compounds
To establish of standard curve of pure compounds,
10 mg of each compound was weighted and
dissolved perfectly in 10 mL MeOH as stock
solution. A certain amount of this solution of each
compound was then taken and diluted to 10 mL in a
volumetric flask to get standard solutions. The
standard curve for each compound was performed as
shown in figures 1 and 2.
Figure 1: Standard curves of (2), (3), (4)
Preparation of sample
100 mg of the ethanol extract was weighted and
dissolved perfectly in 10 mL MeOH then filtered by
using a 0.45-m membrane before injection.
Content calculation of 2-4 in the ethanol extract
The content of each compound in the ethanol
extract was determined by the following formula and
the result was presented in table 1.
C: Concentration of compound (ppm) calculated
from standard curve equation; V: Volumetric
volume (mL); m: Mass of ethanol extract (g);
a: Humidity of ethanol extract (%).
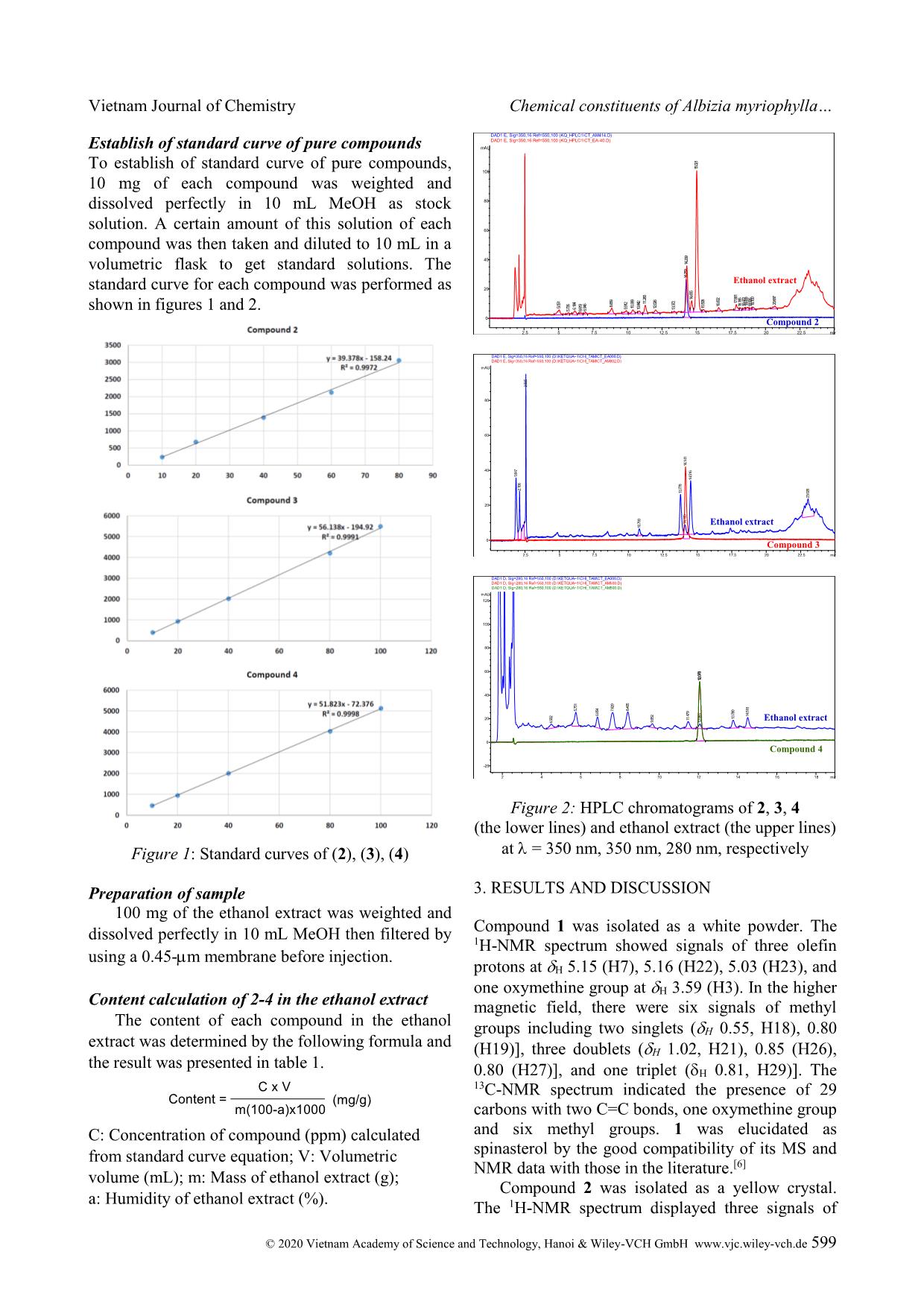
Figure 2: HPLC chromatograms of 2, 3, 4
(the lower lines) and ethanol extract (the upper lines)
at = 350 nm, 350 nm, 280 nm, respectively
3. RESULTS AND DISCUSSION
Compound 1 was isolated as a white powder. The
1H-NMR spectrum showed signals of three olefin
protons at H 5.15 (H7), 5.16 (H22), 5.03 (H23), and
one oxymethine group at H 3.59 (H3). In the higher
magnetic field, there were six signals of methyl
groups including two singlets (H 0.55, H18), 0.80
(H19)], three doublets (H 1.02, H21), 0.85 (H26),
0.80 (H27)], and one triplet (H 0.81, H29)]. The
13C-NMR spectrum indicated the presence of 29
carbons with two C=C bonds, one oxymethine group
and six methyl groups. 1 was elucidated as
spinasterol by the good compatibility of its MS and
NMR data with those in the literature.[6]
Compound 2 was isolated as a yellow crystal.
The 1H-NMR spectrum displayed three signals of
min2.5 5 7.5 10 12.5 15 17.5 20 22.5
mAU
0
20
40
60
80
100
DAD1 E, Sig=350,16 Ref=550,100 (KQ_HPLC1\CT_AM414.D)
1
4.
25
9
DAD1 E, Sig=350,16 Ref=550,100 (KQ_HPLC1\CT_EA-40.D)
5
.0
51
5
.7
36
6
.1
84
6
.6
19
6
.9
46 8
.8
59
9
.9
12
1
0.
38
9
1
0.
84
2
1
1.
28
3
1
2.
02
6
1
3.
37
2
1
4.
29
9
1
4.
65
5
1
5.
02
1
1
5.
50
8
1
6.
60
2
1
7.
88
1
1
8.
18
5
1
8.
47
2
1
8.
65
9
1
8.
92
6
1
9.
10
0
2
0.
68
7
Ethanol extract
Compound 2
min2.5 5 7.5 10 12.5 15 17.5 20 22.5
mAU
0
20
40
60
80
DAD1 E, Sig=350,16 Ref=550,100 (D:\KETQUA~1\CHI_TAM\CT_EA000.D)
1
.8
47
2
.1
06
2
.5
65
1
0.
79
3
1
3.
77
8
1
4.
10
5
1
4.
51
6
2
3.
02
6
DAD1 E, Sig=350,16 Ref=550,100 (D:\KETQUA~1\CHI_TAM\CT_AM802.D)
1
4.
14
1
Ethanol extract
Compound 3
min2 4 6 8 10 12 14 16 18
mAU
-20
0
20
40
60
80
100
120
DAD1 D, Sig=280,16 Ref=550,100 (D:\KETQUA~1\CHI_TAM\CT_EA000.D)
4
.5
02
5
.7
51
6
.8
54 7
.6
29
8
.4
05
9
.6
52
1
1.
47
9
1
2.
08
3
1
3.
78
0
1
4.
51
8
DAD1 D, Sig=280,16 Ref=550,100 (D:\KETQUA~1\CHI_TAM\CT_AM500.D)
1
2.
07
0
DAD1 D, Sig=280,16 Ref=550,100 (D:\KETQUA~1\CHI_TAM\CT_AM500.D)
1
2.
07
0
Compound 4
Ethanol extract
Vietnam Journal of Chemistry Nguyen Thi Hoai Thu et al.
© 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 600
aromatic protons at H 7.95 (1H, d, 8.5 Hz, H5), 6.81
(1H, dd, 8.5, 2.0 Hz, H6), 6.80 (1H, d, 2.0 Hz, H8),
confirming the presence of a 1,2,4-trisubstituted A
benzene ring system of flavonoids. The 1 ,3 ,4 -
trisubstituted B benzene ring was determined by the
signals of three aromatic protons at H 7.60 (1H, d,
1.5 Hz, H2 ), 6.86 (1H, d, 8.5 Hz, H5 ), 7.49 (1H,
dd, 8.5, 1.5 Hz, H6 ). Additionally, the 1H-NMR
spectrum showed one methoxy group at H 3.70. The
13C-NMR spectrum corresponded to 15 carbons,
including one carbonyl carbon at C 175.3 (C4), six
oxygenated aromatic carbons and eight aromatic
carbons (table 2). The HMBC correlation between
protons of -OCH3 with C3 confirmed this methoxy
group attached to C3. Based on all the
aforementioned analysis and the comparison of MS
and NMR data (table 2) with those reported in the
literature,[7] 2 was determined as 7,3',4'-trihydroxy-
3-methoxyflavone.
Table 2: 13C-NMR data of isolated compounds
No
C (ppm)
2a 3b 4a 5b
2 156.1 148.5 80.4 78.5
3 140.1 138.4 44.6 69.4
4 175.3 174.3 192.8 67.2
5 126.9 127.3 129.8 128.3
6 115.1 115.8 111.6 108.3
7 162.5 164.1 165.8 157.4
8 102.3 102.8 103.7 101.9
9 157.2 158.3 164.6 155.1
10 116.9 115.3 114.7 115.0
1 122.5 124.2 131.3 130.4
2 115.3 116.1 114.1 114.0
3 144.5 147.4 145.9 144.6
4 147.6 146.1 145.5 144.6
5 114.9 115.8 115.9 114.6
6 121.4 121.7 119.2 118.0
3-OCH3 59.5 - - -
a) In CDCl3; b) In CD3OD.
Compound 3 was isolated as a yellow crystal.
The NMR data of 3 were similar to those of 2,
except that the methoxy group at C3 was replaced by
a hydroxyl group. Based on the MS and NMR data
(table 2) compared to the ones in published data,[8] 3
was identified as fisetin.
Compound 4 was isolated as a yellow powder.
The NMR data demonstrated that 4 was a flavanone
with the lack of double bond at C2. The comparison
of the measured specific optical rotation value of 4
{[α]D
25 –22.7 (c 0.75, EtOAc)} with the ones of (2S)-
7,3',4'-trihydroxyflavanone isomer {[α]D
22 -36.1 (c
0.8, MeOH) or [α]D
30 -18.7 (c 0.5, MeOH)},[9] as
well as the good compatibility of its MS and NMR
data (table 2) with those published in the
literature,[9,10] 4 was thus (2S)-7,3',4'-
trihydroxyflavanone.
Figure 3: Structures of isolated compounds from
Albizia myriophylla wood
Compound 5 was obtained as a brown powder.
The NMR data suggested that 5 was a flavanane
derived from 3 with the disappearance of the double
bond at C2 and the carbonyl group at C4 and the
appearance of two hydroxyl groups at C3 and C4.
According to Drewes et al.,[11] the coupling
constants J2,3 and J3,4 (in Hertz) of flavan-3,4-diol
derivatives were around 1.0 and 4.4 in (+)-2,3-cis-
3,4-cis isomer; 1.0 and 2.7 in (+)-2,3-cis-3,4-trans
isomer; 10.0 and 3.4 in (+)-2,3-trans-3,4-cis isomer;
and 9.0 and 7.1 in (+)-2,3-trans-3,4-trans isomer,
respectively. Compound 5 possessed a set of proton
signals including 5.02 (brs, H-2), 3.99 (dd, 1.0, 4.0
Hz, H-3) and 4.95 (d, 4.0 Hz) corresponding to the
coupling constants of (+)-2,3-cis-3,4-cis isomer. MS
and NMR data (table 2) of 5 were compatible with
those of published ones.[2,12] The dextrorotatory
value of 5 {[α]D
25 +38.9 (c 4.25, MeOH)} matched
with the one of [α]D
20 +48.5 (c 0.8, acetone-H2O)
reported for (2S,3S,4S)-7,3 ,4 -trihydroxy-2,3-cis-
flavan-3,4-cis-diol.[13] Therefore 5 was assigned as
(2S,3S,4S)-7,3,4,3',4'-pentahydroxyflavane.
The contents of some isolated compounds in the
ethanol extract of A. myriophylla determined by
HPLC were displayed in table 1 and figures 1-2.
From the results, these flavonoids 2-4 were found as
major compounds in the ethanol extract of A.
myriophylla wood. These HPLC chromatograms
could be used to reveal the presence of isolated
compounds as markers of the sample. Fisetin (3) had
Vietnam Journal of Chemistry Chemical constituents of Albizia myriophylla
© 2020 Vietnam Academy of Science and Technology, Hanoi & Wiley-VCH GmbH www.vjc.wiley-vch.de 601
a small content in the ethanol extract, however it
possessed potential interest in the prevention or
treatment of cancer in vivo, antioxidant and anti-
inflammatory[5] The bioactivities of the two rest
flavonoids (2, 4) have not much been studied.
Compound 2 showed no active in assays of the
inhibition of xanthine oxidase[14] as well as
antimalarial activity against Plasmodium
falciparum.[15] Compound 4 was tested for free
radical scavenging activity and its percentage of
reduced DPPH was 14.5 % at a dose of 4 10-8
mol.[15] Therefore, it needs necessary research for the
biological properties of these compounds in order to
support the traditional use of A. myriophylla wood.
4. CONCLUSION
From the Albizia myriophylla wood, collected at Phu
Yen province, spinasterol (1) and four flavonoids (2-
5) were isolated and elucidated the structures. This is
the first time these compounds are known in A.
myriophylla. The contents of some high yield
isolated compounds (2-4) in the ethanol extract were
determined by HPLC and these chromatograms
could be used as markers to verify the presence of
major flavonoids of the sample.
Acknowledgements. We are grateful to Mr. Hoang
Xuan Lam (Co-Founder of HerbEco Company) for
Albizia myriophylla sample and M.Sc. Bui Ngoc Duy
for the good image of the plant.
REFERENCES
1. T. A. T. Nong, Q. T. Nguyen, T. M. T. Nguyen, T. H.
Dao. Study on the phytomorphology of the Albizia
myriophylla Benth. plants collected in Thai Nguyen,
Vietnam J. Sci. Tech., 2017, 17(6), 10-12.
2. N. Joycharat, C. Boonma, S. Thammavong, B.
Yingyongnarongkul, S. Limsuwan, S. P.
Voravuthikunchai. Chemical constituents and
biological activities of Albizia myriophylla wood,
Pharm. Biol., 2016, 54, 62-73.
3. M. Yoshikawa, T. Morikawa, K. Nakano, Y.
Pongpiriyadacha, T. Murakami, H. Matsuda.
Characterization of new sweet triterpene saponins
from Albizia myriophylla, J. Nat. Prod., 2002, 65,
1638-1642.
4. N. Joycharat, S. Thammavong, S. Limsuwan, S.
Homlaead, S. P. Voravuthikunchai, B.
Yingyongnarongkul, S. Dejadisai, S.
Subhadhirasakul. Antibacterial substances from
Albizia myriophylla wood against cariogenic
Streptococcus mutans, Arch. Pharm. Res., 2013, 36,
723-730.
5. Y. S. Touil, N. Auzeil, F. Boulinguez, H. Saighi, A.
Regazzetti, D. Scherman, G. G. Chabot. Fisetin
disposition and metabolism in mice: Identification of
geraldol as an active metabolite, Biochem. Phar.,
2011, 82, 1731-1739.
6. L. J. Goad, Toshihiro Akihisa. Analysis of sterols,
Blackie Academic and Professional, London, UK,
1997, 388.
7. J. H. Wu, Y. T. Tung, S. C. Chien, S. Y. Wang, Y. H.
Kuo, L. F. Shyur, S. T. Chang. Effect of
phytocompounds from the heartwood of Acacia
confusa on inflammatory mediator production, J.
Agri. Food Chem., 2008, 56, 1567-1573.
8. M. P. Costa, M. C. V. Bozinis, W. M. Andrade, C. R.
Costa, A. L. Silva, C. M. Oliveira, L. Kato, O. F.
Fernandes, L. K. H. Souza, M. R. R. Silva.
Antifungal and cytotoxicity activities of the fresh
xylem sap of Hymenaea courbaril L. and its major
constituent fisetin, BMC Comple. Alte. Med. 2014,
14, 245-252.
9. A. R. Jassbi, P. Singh, V. Krishna, P. K. Gupta, S.
Tahara. Antioxidant study and assignments of NMR
spectral data for 3’,4’,7-trihydroxyflavanone 3’,7-di-
O-β-D-glucopyranoside (butrin) and its hydrolyzed
product, Chem. Nat. Com., 2004, 40(3), 250-253.
10. W. L. Kuo, C. C. Chen, P. H. Chang, L. Y. Cheng, B.
J. Shien, Y. L. Huang. Flavonoids from Guizotia
abyssinica, J. Chin. Med., 2007, 18, 121-128.
11. S. E. Drewes, D. G. Roux. Optically active
diastereoisomers of (+)-mollisacacidin by
epimerization, Biochem. J., 1965, 94, 482-487.
12. P. J. Steynberg, J. P. Steynberg, E. V. Brandt, D.
Ferreira, R. W. Hemingway. Oligomeric flavonoids.
Part 26. Structure and synthesis of the first
profisetinidins with epifisetinidol constituent units, J.
Chem. Soc. Perkin Trans., 1997, 28(45), 1943-1950.
13. S. E. Drewes, A. H. Ilsley. Isomeric leucofisetinidins
from Acacia mearnsii, Phytochemistry, 1969, 8(6),
1039-1042.
14. Y. T. Tung, S. T. Chang. Inhibition of xanthine
oxidase by Acacia confusa extracts and their
phytochemicals, J. Agric. Food Chem., 2010, 58,
781-786.
15. M. A. Abdalla, H. Laatsch. Flavonoids from sudanese
Albizia zygia (Leguminosae, subfamily
Mimosoideae), a plant with antimalarial potency, Afr
J. Trad. Comple. Altern Med., 2012, 9(1), 56-58.
Corresponding author: Nguyen Thi Hoai Thu
University of Medicine and Pharmacy at Ho Chi Minh City
217, Hong Bang Street, District 5, Ho Chi Minh City 70000, Viet Nam
E-mail: nguyenthihoaithu@ump.edu.vn; Tel.: +84- 904203342.
File đính kèm:
 a_phytochemical_study_of_the_ethyl_acetate_extract_etoac_of.pdf
a_phytochemical_study_of_the_ethyl_acetate_extract_etoac_of.pdf

